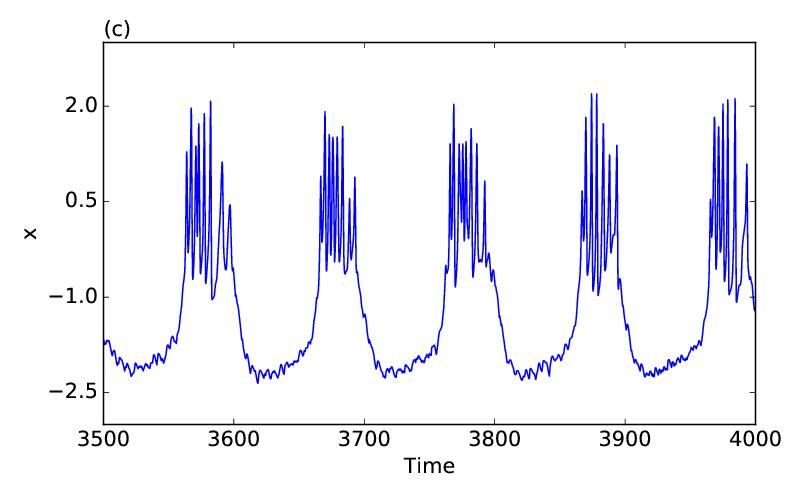
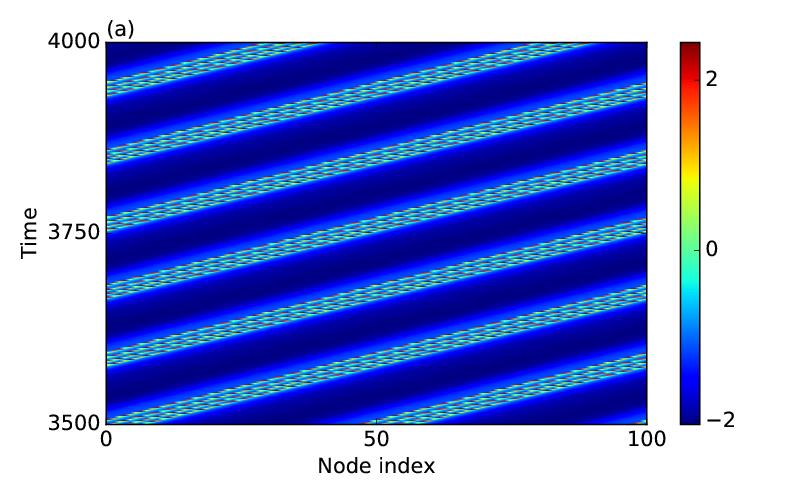
A spatially stable pattern of two coexisting coherent and incoherent subpopulations in nonlocally coupled dynamical systems is called as chimera states and seen in many paradigmatic limit cycle as well as chaotic models where the coupling interaction is basically diffusive type. In neuronal networks, besides diffusive electrotonic communication via gap junctions, chemical transmission occurs between the pre-synapse and post-synapse of neurons. We consider, in a numerical study, a network of neurons in a ring using the Hindmarsh-Rose (HR) bursting model for each node of the network and, apply attractive gap junctions for local coupling between the nearest neighbors and inhibitory nonlocal coupling via chemical synaptic transmission between the distant neighbors. For a range of gap junctional and chemical synaptic coupling strengths, a subpopulation of the neuronal network, in the ring, bursts asynchronously and another subpopulation remains silent in a synchronous state. The bursting subpopulation of neurons fires sequentially along the ring when the number of firing nodes remains same but change their positions periodically in time. It appears as a traveling chimera pattern in the ring when the dynamics of the individual bursting nodes is chaotic. The chimera pattern travels in a reverse direction for a larger chemical synaptic coupling strength. A purely inhibitory chemical synaptic coupling can produce a similar traveling chimera pattern, however, the dynamics of the firing nodes is then periodic.

| Attachment | Size |
|---|---|
| 1.12 MB |
A.M is supported by the University Grants Commission, India. A.M. and S.S also acknowledge local support and hospitality for two week’s visit by the Lobachevsky state University of Nizhny Novgorod, Russia. D.G. and S.K.D. are supported by the SERB-DST (Department of Science and Technology), Government of India (Project No. INT/RUS/RFBR/P-181), GO acknowledges support of Russian Science Foundation (Project N 17-12-01534).
Abrams, D. M., Mirollo, R., Strogatz, S. H., and Wiley, D. A. (2008). Solvable model for chimera states of coupled oscillators. Phys. Rev. Lett., 101:084103.
Abrams, D. M. and Strogatz, S. H. (2004). Chimera states for coupled oscillators. Phys. Rev. Lett., 93:174102.
Bera, B. K. and Ghosh, D. (2016). Chimera states in purely local delay-coupled oscillators. Phys. Rev. E, 93:052223.
Bera, B. K., Ghosh, D., and Banerjee, T. (2016a). Imperfect traveling chimera states induced by local synaptic gradient coupling. Phys. Rev. E, 94:012215.
Bera, B. K., Ghosh, D., and Lakshmanan, M. (2016b). Chimera states in bursting neurons. Phys. Rev. E, 93:012205.
Bressler, S. L. and Kelso, J. (2001). Cortical coordination dynamics and cognition. Trends in Cognitive Sciences, 5(1):26 – 36.
Dudkowski, D., Maistrenko, Y., and Kapitaniak, T. (2014). Different types of chimera states: An interplay between spatial and dynamical chaos. Phys. Rev. E, 90:032920.
Friston, K. J. (1997). Transients, metastability, and neu-ronal dynamics. NeuroImage, 5(2):164 – 171.
Glaze, T. A., Lewis, S., and Bahar, S. (2016). Chimera states in a hodgkin-huxley model of thermally sensitive neurons. Chaos: An Interdisciplinary Journal of Nonlinear Science, 26(8):083119.
Gu, C., St-Yves, G., and Davidsen, J. (2013). Spiral wave chimeras in complex oscillatory and chaotic systems. Phys. Rev. Lett., 111:134101.
Hagerstrom, A. M., Murphy, T. E., Roy, R., Hovel, P., Omelchenko, I., and Scholl, E. (2012). Experimental observation of chimeras in coupled-map lattices. Nature Physics, 8:658.
Hens, C. R., Mishra, A., Roy, P. K., Sen, A., and Dana, S. K. (2015). Chimera states in a population of identical oscillators under planar cross-coupling. Pramana – Journal of Physics, 84:229.
Hizanidis, J., Kanas, V. G., Bezerianos, A., and Bountis, T. (2014). Chimera states in networks of nonlocally coupled hindmarsh–rose neuron mod-els. International Journal of Bifurcation and Chaos, 24(03):1450030.
Kaneko, K. (1990). Clustering, coding, switching, hi-erarchical ordering, and control in a network of chaotic elements. Physica D: Nonlinear Phenomena, 41(2):137 – 172.
Kuramoto, Y. and Battogtokh, D. (2002). Coexistence of coherence and incoherence in nonlocally coupled phase oscillators. Nonlinear Phenomena in Complex Systems, 5:380–385.
Larger, L., Penkovsky, B., and Maistrenko, Y. (2013). Virtual chimera states for delayed-feedback systems. Phys. Rev. Lett., 111:054103.
Lazarides, N., Neofotistos, G., and Tsironis, G. P. (2015). Chimeras in squid metamaterials. Phys. Rev. B, 91:054303.
Levanova, T. A., Komarov, M. A., and Osipov, G. V. (2013). Sequential activity and multistability in an ensemble of coupled van der pol oscillators. The Eu-ropean Physical Journal Special Topics, 222(10):2417– 2428.
Levy, R., Hutchison, W. D., Lozano, A. M., and Dostro-vsky, J. O. (2000). High-frequency synchronization of neuronal activity in the subthalamic nucleus of parkinsonian patients with limb tremor. Journal of Neuroscience, 20(20):7766–7775.
Majhi, S., Perc, M., and Ghosh, D. (2016). Chimera states in uncoupled neurons induced by a multi-layer structure. Scientific Reports, 6:39033.
Maksimenko, V. A., Makarov, V. V., Bera, B. K., Ghosh, D., Dana, S. K., Goremyko, M. V., Frolov, N. S., Ko-ronovskii, A. A., and Hramov, A. E. (2016). Excitation and suppression of chimera states by multiplexing. Phys. Rev. E, 94:052205.
Martens, E. A., Laing, C. R., and Strogatz, S. H. (2010). Solvable model of spiral wave chimeras. Phys. Rev. Lett., 104:044101.
Martens, E. A., Thutupalli, S., Fourriere,` A., and Hal-latschek, O. (2013). Chimera states in mechanical oscillator networks. Proceedings of the National Academy of Sciences, 110(26):10563–10567.
Mathews, C. G., Lesku, J. A., Lima, S. L., and Am-laner, C. J. (2006). Asynchronous eye closure as an anti-predator behavior in the western fence lizard (sceloporusoccidentalis). Ethology, 112(3):286–292.
Mikhaylov, A. O., Komarov, M. A., Levanova, T. A., and Osipov, G. V. (2013). Sequential switching activity in ensembles of inhibitory coupled oscillators. EPL (Europhysics Letters), 101(2):20009.
Mishra, A., Hens, C., Bose, M., Roy, P. K., and Dana, S. K. (2015). Chimeralike states in a network of oscillators under attractive and repulsive global coupling. Phys. Rev. E, 92:062920.
Mishra, A., Saha, S., Hens, C., Roy, P. K., Bose, M., Louodop, P., Cerdeira, H. A., and Dana, S. K. (2017a). Coherent libration to coherent rotational dynamics via chimeralike states and clustering in a josephson junction array. Phys. Rev. E, 95:010201.
Mishra, A., Saha, S., Roy, P. K., Kapitaniak, T., and Dana, S. K. (2017b). Multicluster oscillation death and chimeralike states in globally coupled joseph-son junctions. Chaos: An Interdisciplinary Journal of Nonlinear Science, 27(2):023110.
Omelchenko, I., Maistrenko, Y., Hovel,¨ P., and Scholl,¨ E. (2011). Loss of coherence in dynamical networks: Spatial chaos and chimera states. Phys. Rev. Lett., 106:234102.
Rattenborg, N. C., Amlaner, C. J., and Lima, S. L. (2000). Behavioral, neurophysiological and evolu-tionary perspectives on unihemispheric sleep. Neuroscience &Biobehavioral Reviews, 24(8):817 – 842.
Rattenborg, N. C., Lima, S. L., and Amlaner, C. J. (1999). Half-awake to the risk of predation. Nature, 397:397–398.
Schmidt, L. and Krischer, K. (2015). Clustering as a prerequisite for chimera states in globally coupled systems. Phys. Rev. Lett., 114:034101.
Wickramasinghe, M. and Kiss, I. Z. (2013). Spatially organized dynamical states in chemical oscillator networks: Synchronization, dynamical differentiation, and chimera patterns. PLOS ONE, 8(11).
Yeldesbay, A., Pikovsky, A., and Rosenblum, M. (2014). Chimeralike states in an ensemble of globally coupled oscillators. Phys. Rev. Lett., 112:144103.