Introduction
The ability of the mammalian nervous system to detect and discriminate external and internal temperatures is crucial for the homeostasis and survival. Temperature related changes in neurons excitability provide this ability. With discovery of TRP channels specifically sensitive to temperature (thermoTRPs), changes in neuronal excitability became attributed almost exclusively to these channels as they are activated in physiologically important range (0–60 °C) and conduct depolarizing currents. In recent years, the functional role of temperature dependence of other type ion channels is revisited (Vriens, Nilius, & Voets, 2014). Whereas the expression, biophysical properties, and functional roles of the thermoTRPs are matters of numerous reviews (Michael J Caterina & Pang, 2016; Talavera, Nilius, & Voets, 2008; Voets, 2012), the temperature-related properties of the non-TRP channels are not systematically reviewed. Here we (1) consider which and how biophysical properties of non-TRP channels depend on temperature and (2) elucidate how this temperature-dependence influences the neuronal excitability and, consequently, some physiological functions. The scope of this review is limited to the data obtained in warm-blooded animals. We address the following specific questions. Which non-TRP channels significantly change their biophysical properties with temperature? Which specific properties of the channels conducting de- and hyperpolarizing currents are influenced by temperature? In which ways these influences are performed? What are possible functional consequences of normal or altered thermosensitivity of the non-TRP channels in health and disease?
Voltage-gated channels sensitive to temperature
Channels conducting depolarization currents
Ca2+ channels
Temperature sensitivity of the biophysical characteristics is described for Cav3.1-3.3 also known as low-voltage activated (LVA), T-type and Cav 1.2 and Cav 1.3, high-voltage activated (HVA), L-type (Chemin et al., 2007; Coulter, Huguenard, & Prince, 1989; Iftinca et al., 2006; Johnson & Marcotti, 2008; Narahashi, Tsunoo, & Yoshii, 1987; Nobile, Carbone, Lux, & Zucker, 1990; Radzicki et al., 2013; Rosen, 1996).
Na+ channels
Temperature dependence was found in TTX-sensitive (TTXs, subtypes Nav 1.1 – 1.4; Nav 1.6; Nav 1.7) and TTX-resistant (TTXr, subtypes Nav 1.5; Nav 1.8; and Nav 1.9) sodium channels (Rosen, 2001; Sarria, Ling, & Gu, 2012; Zimmermann et al., 2007). The Nav1.1, Nav1.6, and Nav1.7 channels are also called transient sodium (NaT) channels because of their fast activating and inactivating kinetics. Correspondingly, the non-inactivating or slowly inactivating channels of Nav1.8 and Nav1.9 subtypes are termed persistent sodium (NaP) channels. Occurrence of pathological sensitivity to temperature is described for Nav1.2 with mutated β1-subuinit (Egri, Vilin, & Ruben, 2012).
HCN channels
Strong temperature dependence was demonstrated for hyperpolarization-activated channels conducting cationic depolarizing currents of HNC1-type influencing the discharge patterns of mammalian cold thermoreceptor endings. Pathological thermosensitivity occurring in mutated HCN2 was shown to contribute to febrile seizures (Nakamura et al., 2013).
Channels conducting hyperpolarization currents
K+ channels
Temperature sensitivity was described for channels conducting A-type potassium currents (Sarria et al., 2012), for the Kv2.1 and Kv4.3 channels with uncoupled voltage sensor (Yang & Zheng, 2014), and for the TREK-1, TREK-2, and TRAAK two-pore channels (Schneider, Anderson, Gracheva, & Bagriantsev, 2014).
Cl− channels
Highly temperature dependent properties are inherent in the anoctamin-1 (ANO1, also known as TMEM16) Ca2+-activated chloride conducting channels (Cho et al., 2012) and CLC proteins mediating Cl− transfer (Pusch & Zifarelli, 2014).
Biophysical properties altered by temperature
Peak and steady currents, current-voltage relations
Whole cell voltage-clamp recordings at 21 °C and 37 °C from tsA-201 cells exogenously expressing the three different rat brain T-type calcium channel isoforms, Cav3.1, Cav3.2, and Cav3.3 and the human isoform, Cav3.3h showed that the heating led to increase in Ca2+ current amplitude by 1.8-fold for Cav3.1, 2.1-fold for Cav3.2, 1.6-fold for Cav3.3, and 1.8-fold for Cav3.3h (Iftinca et al., 2006). The effects were the same when temperature was either ramped up from 21 °C, or down from 37 °C. Calcium current of L-type (predominantly Cav1.3) recorded from the inner hear cells (IHCs) of immature (<P12) and adult (>P20) gerbils at body temperature (35–37 °C) was significantly greater in magnitude than that recorded at room temperature (21–23 °C) with the temperature ratios Q10 =1.9 and 1.4 for immature and adult animals, respectively. Similar temperature dependence was observed for the steady current through L-type (Cav1.3) channels in IHCs of 2-week-old C57Bl6/N mice (Nouvian, 2007) with the temperature ratios of Q10 =1.1 (whole-cell recordings) or Q10 =1.3 (perforated patch). The significantly steeper temperature dependence of this current in immature IHCs suggests existence of developmental change in these properties. Based on the singe-channel recordings from dissociated dorsal root ganglion (DRG) neurons of 12-day old chicken embryo, it is suggested that the larger Ca2+ current at physiological temperature is likely due to an increased opening probability (Acerbo & Nobile, 1994).
Cooling from 30 °C to 10 °C reduced peak values of both TTX-sensitive and TTX-resistant sodium currents (through channels of Nav1.7 and Nav1.8 subtypes, respectively), recorded in cultured small-size DRG neurons of rat and heterologous cells expressing these channels (Sarria et al., 2012; Zimmermann et al., 2007). The decrease in current amplitudes on cooling was significantly stronger for Nav1.7 than for Nav1.8, especially at a membrane potential of -80 mV, which is similar to the physiological membrane potential. Study of sodium current through TTXs channels of GH3 cells from rat pituitary showed absence of noticeable change in the current-voltage relationship with temperature changing in the range from 23 to 37 °C (Rosen, 2001). In Chinese Hamster Ovary (CHO) cells expressing Nav1.2, particularly with beta1 subunit, it was found that elevation of temperature from 22 to 34 °C causes increase in the persistent and window current (Egri et al., 2012).
A-type K+ currents (IA) were strongly inhibited by cooling, whereas non-inactivating K+ currents (IK) were weakly inhibited by this cue (Sarria et al., 2012). Experiments on HEK293 cells expressing Kv2.1 and Kv4.3 channels with the use of the voltage ramp protocol (−100 to +50 mV in 300 ms) revealed a large increase in peak currents through those channels with heating from 21 °C to 33 °C (Kv2.1) or 23 °C to 39 °C (Kv4.3) (Yang & Zheng, 2014). This was observed within a relatively narrow voltage range (near – 50 mV). In this range the heating-induced increase in Kv2.1 and Kv4.3 conductance was characterized by a transient increase in Q10 to very high values of about 20 to 30 that is more characteristic of specialized thermosensitive channels of TRP family. At normal body temperature, the two-pore potassium (K2P) channels TREK-1 (K2P2.1/KCNK2), TREK-2 (K2P10.1/KCNK10), and TRAAK (K2P4.1/KCNK2) regulate cellular excitability by providing voltage-independent leak of potassium. Heat dramatically potentiates K2P channel activity and further affects excitation (Schneider et al., 2014). At room temperature, TREK-1 exhibits only background potassium leak, which increases with temperature, reaching maximum at ~42 °C. Interestingly, TREK-1 has its half-maximal temperature activation point (T1/2) at ~37 °C, implying that the midpoint of the channel's dynamic range is centered on the homeostatic thermal set point for most mammals.
Chloride current through ANO1 increased by heating with a steeper temperature dependence at higher intracellular Ca2+ concentrations (Cho et al., 2012). Notably, raising the temperature above 44 °C resulted in robust inward Cl− currents at a holding potential of −60 mV in ANO1-HEK cells. Heat-evoked ANO1 currents in these cells were outwardly rectifying and reversed at near zero mV (−5.0 mV, n = 5).
Voltage-dependence of activation and inactivation
In the above mentioned experiments on Cav3.3 isoforms (Iftinca et al., 2006), heating from 21 °C and 37 °C caused a small (~9 mV) but statistically significant hyperpolarizing shift of the voltage-dependent activation for the Cav3.1 (the half-activation potentials Va1/2 changed from –52.4±0.7 to –60.5±0.9 mV) and Cav3.2 (from –42.9±0.8 to –51.5±1.0 mV); however, the shift was insignificant for rat Cav3.3 (from –72.9±1.1 to –73.5±1.3 mV) or human Cav3.3h (from –55.5±1.1 to –56.8±0.9 mV). The temperature ratio (Q) of the Va1/2 values calculated at the two temperatures for Cav3.1 and Cav3.2 was Q=1.15 and 1.2, respectively. Neither isoform displayed significant change in the slope of its activation voltage-dependence. In the same experiments, heating also shifted the voltage-dependent inactivation. There was a hyperpolarization shift in half-inactivation potential Vh1/2 for Cav3.1 (from –72.2±1.1 to –75.4±4.3 mV), Cav3.2 (from –64.2±2.2 to –73.7±1.2 mV), and Cav3.3h (from –69.0±1.9 to –82.1±1.5 mV) and a small depolarizing shift for the rat Cav3.3 (from –78.3±0.8 to –73.4±2.5 mV). The effects were statistically significant only for the Cav3.2 subtype and Cav3.3h (with temperature ratios Q=1.15 and 1.19, respectively). The combined effects of temperature on half activation and half inactivation potentials further resulted in a hyperpolarizing shift in the position of the window current (i.e. the overlap between activation and inactivation curves) for Cav3.1 and Cav3.2, which would allow these channels to be active at neuronal resting membrane potentials (Perez-Reyes, 2003).
Sodium currents, both TTX-sensitive (TTXs) and TTX-resistant, also displayed a small shift of the voltage-dependent activation towards more hyperpolarized potentials, however this was caused by cooling from ~30 to 10°C. This was observed in the mentioned above experiments on cultured DRG neurons expressing Nav1.7 (TTXs) and Nav1.8 (TTXr) channels (Zimmermann et al., 2007). In the same experiments, cooling had little effect on fast inactivation but markedly shifted the slow inactivation of TTXs currents towards more hyperpolarized potentials. In contrast, slow inactivation of TTXr currents was resistant to cooling. These distinct properties of TTXs and TTXr currents were conserved in recombinant Nav1.7 (TTXs) and Nav1.8 (TTXr) channels; that is, cooling resulted in a leftward shift of slow inactivation of Nav1.7 but not of Nav1.8 (Zimmermann et al., 2007). A consistent result was obtained in other studies on rat DRG neurons (Sarria et al., 2012), in which steady-state slow inactivation curve of TTXs channel shifted to more hyperpolarizing voltages with cooling that resulted in substantial slow inactivation near resting membrane potentials. In CHO cells expressing Nav1.2 β1 subunit, it was found that elevation of temperature from 22 to 34°C causes a hyperpolarization shift of the voltage-dependent activation for all subunits (Egri et al., 2012). The same effect was found for the fast inactivation. The rate of fast inactivation recovery was also increased by elevated temperature for all subunits.
The above mentioned heat-induced augmentation of potassium currents through Kv2.1 and Kv4.3 channels was not due to a shift of the half-activation potential V1/2 as the peak Q10 values exhibited no correlation with the temperature-dependent shifts in V1/2 (Yang & Zheng, 2014).
Rates (time constants) of voltage-dependent activation and inactivation
Increasing the temperature from 21 °C to 37 °C led to a large and statistically significant reduction in the time constants of both activation and inactivation for rat Cav3.1, Cav3.2, Cav3.3 and human Cav3.3h isoforms of Cav3 channels explored in the above-mentioned study (Iftinca et al., 2006). The accelerating effect of such heating on the inactivation kinetics of Cav3.1 and Cav 3.3 was apparent in a wide range of voltages, but in case of Cav 3.2 it was observed only below –20 mV. Noteworthy, the inactivation time course was well described by a single exponential at 21 °C whereas at 37 °C it became bi-exponential because of occurrence of a second slower decay component. Absence of the latter component at 21 °C is particularly notable as it has often been considered as a feature of this T-type channel isoform. Nonetheless, Cav3.3 channels still inactivated more slowly than the other T-type channel subtypes (Iftinca et al., 2006). In view of its significance for duration of the refractory period after neuron APs termination, the temperature dependence of the time required for recovery from inactivation is of particular interest. The time constant of this process significantly decreased with heating from 21 °C to 37 °C as follows from measurements performed at –120 mV for Cav3.1 (Q=0.4), Cav3.2 (Q=0.35), rat Cav3.3 (Q=0.33), and human Cav3.3h (Q=0.3). At 37 °C, the recovery times were similar for all three isoforms suggesting their similar ability to recover from the inactivated state at physiological temperatures. Recovery from inactivation examined at other membrane potentials (–80 and –100 mV for Cav3.1 and Cav3.2; and –90 and –110 mV for Cav3.3) was also significantly accelerated at 37 °C irrespective of membrane potential. However, unlike at more negative recovery potentials, at –80 mV Cav3.2 recovered from inactivation more slowly than Cav3.1. At 21 °C and 37 °C, the recovery time constants for Cav3.1 were 444.1 ±76.2 ms and 197.3 ±15.5 ms, respectively (Q=0.44) and for Cav3.2 they were 722.9 ±73.0 ms and 488.7 ±48.7 ms, respectively (Q=0.68). The recovery could not be examined for rat Cav3.3 at –80 mV because of its more negative activation and inactivation range. For the same three isoforms of Cav3 channels, time constants of deactivation were determined from the tail currents recorded at 21 °C at 37 °C and fitted with single exponential functions. At both temperatures, they were similar for Cav3.2 regardless of the membrane potentials (–120, –100, and –80 mV) and became significantly different at more depolarized potentials for Cav3.1 and rat Cav3.3 (Fig. 4B, D). Interestingly, at both temperatures the rat and human Cav3.3 channel showed the slowest deactivation kinetics compared with the other two isoforms irrespective of the holding potential, suggesting that this channel subtype may have an energetically more stable open conformation. At 37 °C, the deactivation time constants were comparable for the three rat isoforms at potentials more negative than –100 mV, but rat Cav3.3 was substantially slower to deactivate at typical neuronal resting potentials. Moreover, the values obtained for Cav3.3h channels were smaller than those seen with the rat Cav3.3 isoforms, but still showed temperature dependence (Q=1.15 for Cav3.1; Q=0.83 for Cav3.2; Q=0.56 for Cav3.3; and Q=0.42 for Cav3.3h). Studies of calcium L-type (Cav1.3) current in mouse IHCs showed that with lowering the temperature from~37 °C to room temperature the activation time constant was slowed down by about half (Marcotti, Johnson, Rusch, & Kros, 2003; Nouvian, 2007). Measured at -20 mV average time constant of activation τm decreased (activation accelerated) with temperature increase in the range from 23 °C to 37 °C with sharper slope (greater temperature sensitivity) in the lower temperature sub-range 23-28 °C compared to higher temperature sub-range. This feature was especially evident at lower membrane voltages. The inactivation time constant τh of this current linearly decreased (inactivation accelerated too) in the whole mentioned temperature range (Egri et al., 2012; Rosen, 2001).
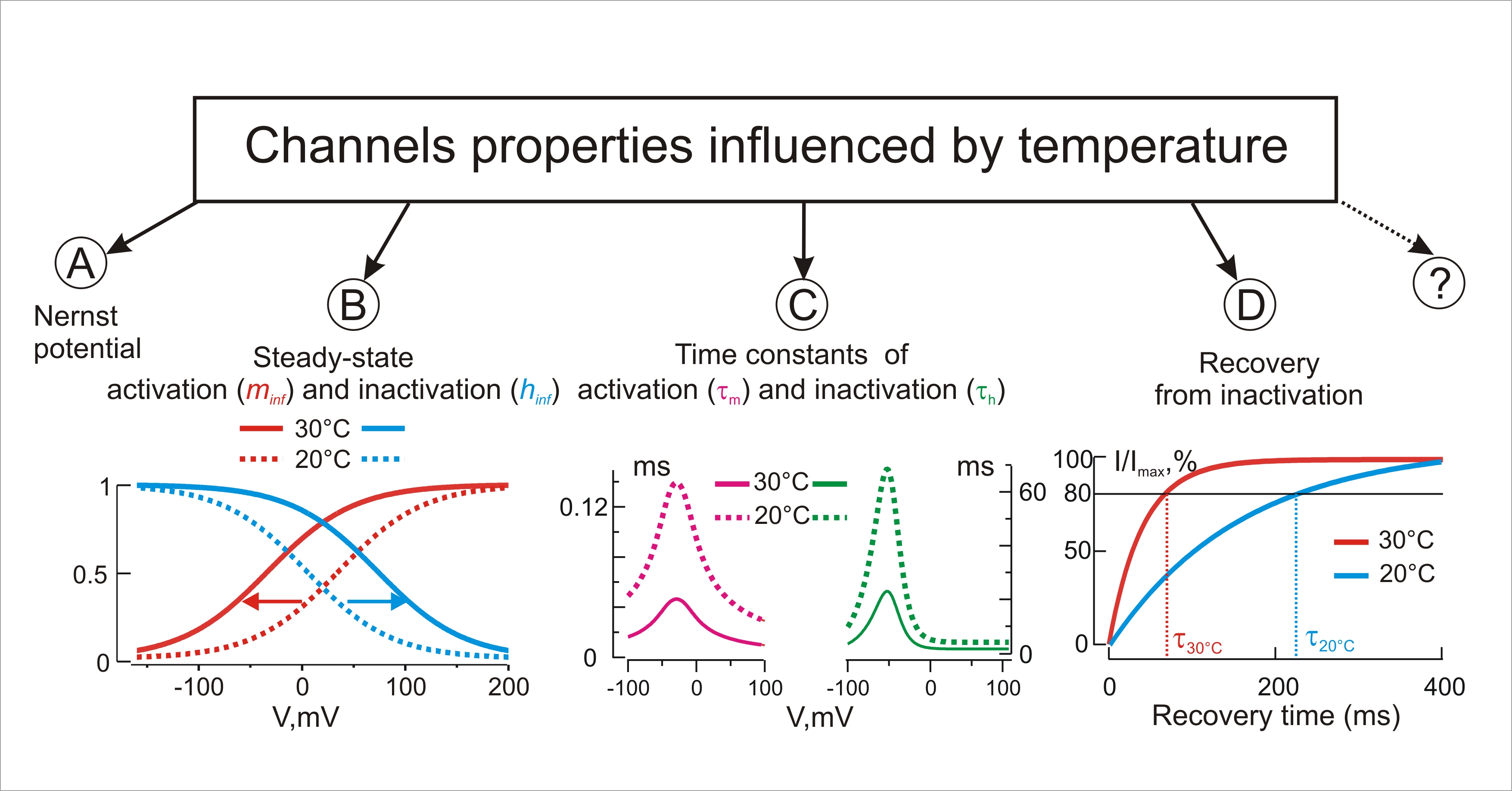
Figure 1. Temperature influences the Nernst equilibrium potential (A), steady-state kinetics (B) and time constants (C) of activation and inactivation, recovery from inactivation (D), and, possibly, other yet unknown biophysical properties of channels.
Functional implications of channels thermosensitive properties in health and disease
Temperature dependence is inherent in practically all type ion currents as corresponding current-conducting channels change their biophysical properties with temperature. These changes are quantitatively characterized by the temperature coefficient Q10. It can be calculated as the relative increase in current amplitude at a specific voltage when temperature increases by 10 degrees, i.e. as Q10 = IT+10/IT. Q10≥5 was proposed as a criterion for distinguishing thermosensitive TRP channels (thermoTRPs) from the whole TRP family (Voets, 2012). According this criterion some non-TRP channels also can be have qualified as highly thermosensitive as they have Q10≥5 (e.g. CLC , ANO1 (Pusch & Zifarelli, 2014). Unlike channels, there is no generally admitted criterion for distinguishing a thermosensory unit at the level of individual neurons. We consider that expression of channels with so high Q10 by itself is necessary but not sufficient for a neuron to function as a thermosensory unit. Our suggestion is supported by known expression of highly thermosensitive TRPV1 and TRPM8 channels in neurons, which do not function as thermosensors. Expression of the high-Q10 channels must be sufficiently high for providing a steep temperature dependence of a neuron reaction (e.g. firing rate) to changes in temperature. Neurons, which are not high-thermosensitive in health can become such in result of channelopathy that leads to development and maintenance of pathological states (febrile seizures). However, implications of existing temperature dependence of CLC proteins, which mediate Cl− transport, are unknown (Pusch & Zifarelli, 2014). This is also true in case of mechanosensitive Piezo-1 and Piezo-2 channels clearly displaying temperature dependence (Coste et al., 2010).
Physiological functions
Central thermodetection
Participation of TRP channels in thermoregulatory responses of neurons in the preoptic area and anterior hypothalamus (PO/AH) is disputed. On the one hand, mechanism for PO/AH neuronal warm sensitivity was attributed to thermosensitive TRPV4 channels (Caterina et al., 1997; Güler et al., 2002; Patapoutian, Peier, Story, & Viswanath, 2003). On the other hand, Wechselberger and co-authors (Wechselberger, Wright, Bishop, & Boulant, 2006) showed the absence of TRPV4 channels expression in PO/AH neurons and, in the simulation study, they demonstrated that the potassium leak and hyperpolarization-activated cationic channels (HCN) determined warm-induced responses of PO/AH neurons. Recent study (Kamm & Siemens, 2017) demonstrated that other type TRP-channel, namely TRPM2 is involved in thermoregulatory responses of PO/AH neurons. They showed that heat (>37 °C) responses of POA neurons were significantly reduced in TRPM2-deficient mice compared to wild-type ones. Two-pore potassium channels, namely TREK are also involved in thermoregulatory responses of neurons in PO/AH. TREK are activated by heating and thus inhibit depolarization of these PO/AH neurons (Maingret et al., 2000).
Peripheral thermodetection
Cold reception. It is generally thought that activity of TRPM8 and TRPA1 channels underlies sensitivity of PANs to innocuous and noxious cold, respectively, while the role of other channels is underestimated. For instance, in cold nociceptor neurons, cooling progressively enhances the voltage-dependent slow inactivation of TTX-sensitive sodium channels, whereas the inactivation properties of TTX-insensitive Nav1.8 channels are entirely cold-resistant. The observation that Nav1.8-deficient mice show negligible responses to noxious cold and mechanical stimulation at low temperatures suggests that Nav1.8 remains available as the sole electrical impulse generator in nociceptors that transmits nociceptive information to CNS (Zimmermann et al., 2007). Decreased activation of TREK-1 potassium channels at low temperatures reduces their contribution of the membrane hyperpolarization and thus promotes the depolarization of receptor endings of cold PANs (Maingret et al., 2000).
Warm reception. Activity of vast majority of ion channels is increased and/or accelerated with increasing temperature. This was shown for practically all type channels conducting the depolarizing currents. In case of the hyperpolarizing current conducting channels this property was demonstrated only for the two-pore potassium (K2P) ones. Moreover, K2Ps may lose selectivity to potassium under physiologically relevant conditions that may turn them into a heat-activated excitatory ion channel similar to TRPV1, i.e. its own functional antipode. In PANs from TRV1-KO mice, APs generation in response to ramp-heating was attributed to presence of high temperature-sensitive (Q10 ~20) Ca2+ activated chloride channels of ANO1-type (Cho et al., 2012).
Role in neuropathology
The role of ion channels of other than thermoTRP types in temperature dependence of pathological processes was shown for development and treatment of neuronal hyperexcitability. On the one hand, NaP with mutated β1 (C121W) subunit shows high temperature sensitivity by widening window current which is one of the possible mechanism of febrile seizures (Egri et al., 2012). On the other hand, it’s known that therapeutic hypothermia suppresses excessive excitability of cortical neurons involved in generation of epileptiform EEG patterns (Motamedi, Gonzalez-Sulser, Dzakpasu, & Vicini, 2012). In our recent model study (Demianenko, Poddubnaya, Makedonsky, Kulagina, & Korogod, 2017) we demonstrated cooling induced deactivation of termoTRP can be a mechanism of suppression of bursting activity of neurons underlying synchronized epileptiform network activity. But role of other non- thermoTRPs is not sufficiently disclosed until now. Heat, voltage, and Ca2+ synergistically sensitize each other’s effects on ANO1. Normally, ANO1 are activated by noxious heat. However, temperatures lower than 44 °C also can activate ANO1 at physiological membrane potentials because [Ca2+]i increases under pathological conditions such as inflammation (Cho et al., 2012).







