Astrocytes perform fundamental housekeeping functions in the central nervous system and through bidirectional communication with neurons are thought to coordinate synaptic transmission and plasticity. They are also renowned actors in brain pathology. Reactive gliosis and neuroinflammation are featured by many (if not all) acute and chronic neurodegenerative pathologies including Alzheimer’s disease (AD). The Ca2+/calmodulin-activated phosphatase calcineurin (CaN) plays a central role in the pathology-related changes of astroglial cells mainly through activation of the inflammation-related transcription factors Nuclear Factor of Activated T-cells (NFAT) and Nuclear Factor kB (NF-kB). In this contribution we focus on the mechanistic aspects of CaN signalling in astrocytes. We analyze the astroglial Ca2+ signalling toolkit in the context of Ca2+ signals necessary for CaN activation and focus on the astroglial CaN signalling through its direct target, NFAT, as well as the intricate relationships between CaN and NF-kB activation pathways.The majority of data about CaN-mediated signalling in astrocytes point to the role for CaN in pathology-related conditions while very little is currently known about signalling and function of astroglial CaN in physiology.
Introduction
Calcineurin (CaN), a Ca2+/calmodulin (CaM)-activated serine/threonine phosphatase, serves as a master-regulator of development- and differentiation-associated cellular remodelling. CaN signalling is mostly studied in peripheral immune cells (Fric et al., 2012; Pan et al., 2013), muscle cells(Mallinson et al., 2009; Schiaffino, 2010; Hudson & Price, 2013), and developing neurons (Carafoli et al., 1999; Genazzani et al., 1999; Kramer et al., 2003). In differentiated neurons, changes of CaN activity are essential for establishment of long-term depression (Mulkey et al., 1994) and emotional memory (Baumgärtel et al., 2008; Baumgärtel & Mansuy, 2012), maintenance of neuronal excitability and metaplasticity (Abraham & Bear, 1996). In neuronal pathology, increased CaN signalling is implicated both in neuroinflammation and in memory impairment, the latter being likely to be caused by a combination of factors including decreased synaptic plasticity, synaptic loss, neuronal apoptosis and neuroinflammation (Reese & Taglialatela, 2010; Reese & Taglialatela, 2011). Regarding astroglial cells, CaN signalling is studied principally in pathological conditions (Reese & Taglialatela, 2010; Reese & Taglialatela, 2011; Furman & Norris, 2014) in most of which, including Alzheimer’s disease (AD), the augmented expression/hyperactivation of CaN was found. Activation of CaN is implicated in disease-associated inflammation and reactive astrogliosis both in chronic neurodegeneration (Furman & Norris, 2014) and in acute brain pathology (Kaminska et al., 2004). Neuroinflammation is a complex polyfactorial phenomenon in which astrocytes share the centre stage with microglial cells (Heneka et al., 2015; Heppner et al., 2015; DiSabato et al., 2016). The role of astrocytes in such an inflammatory duet, in which astrocytes can exert both neuroprotective and neurotoxic effects, is not yet precisely defined and is a matter of intensive investigations(Pekny & Pekna, 2014; Ben Haim et al., 2015). Astroglial CaN, in this context, acts as a molecular switch for setting-up astroglial reactivity and for amplification of inflammatory cascades (Schubert et al., 2000; Furman & Norris, 2014), yet it is required for the resolution of inflammation (Fernandez et al., 2007; Fernandez et al., 2012). Inflammatory aspects of CaN signalling in astrocytes have been recently reviewed in several comprehensive contributions addressing different aspects of this complex issue (Reese & Taglialatela, 2010; Reese & Taglialatela, 2011; Furman & Norris, 2014).
The present contribution is focused on the mechanistic aspects of CaN signalling in astrocytes. We analyse the literature reporting the Ca2+ signals, necessary for the activation of CaN and its downstream targets, and the signalling in astrocytesmediated by the direct CaN target, NFAT, as well as complex and intricate relationships with the signalling mediated by NF-kB.
Calcineurin
Molecular principles of CaN signalling
Among the plethora of processes and enzymes at work in the cell, which are regulated by spatio-temporally defined changes in intracellular Ca2+ concentration ([Ca2+]i), the serine/threonine phosphatase CaN is the only phosphatase directly activated by Ca2+ and CaM binding. This unique feature places CaN on the apex of Ca2+ regulated signalling cascades which require protein dephosphorylation, and it is difficult to overestimate its role in cellular (patho)physiology. In the field of brain pathology, there is growing interest for CaN as it is thought to be the main promoter of neuroinflammation and of transition to reactivity in astrocytes. Before discussing CaN signalling in astrocytes, we shall briefly describe the molecular principles of CaN activation and the modulation of its activity by Ca2+ signals.
CaN was discovered around four decades ago as an inhibitor of the CaM-dependent cyclic nucleotide phosphodiesterase (Wang & Desai, 1976; Klee et al., 1979). It was first characterized in neural tissue and then was found as a largely distributed protein in all tissues. Structurally, CaN is a heterodimer of a ̴60 kDa catalytic A subunit (also known as calcineurin A or CaNA) and it is tightly associated with a ̴19 kDa regulatory B subunit (also known as calcineurin B or CaNB). CaNA possesses the catalytic site at the N-terminal portion. In addition it has two binding domains, one recognized by CaNB and the other one able to bind CaM. In mammals three isoforms of CaNA have been identified: α, β and γ(Goto et al., 1986a; Klee et al., 1988; Kuno et al., 1992; Cohen et al., 1996). The isoforms α and β are ubiquitously distributed, while the γ isoform is specifically expressed in testis (Muramatsu & Kincaid, 1992).
The CaNB subunit possesses four distinct helix-loop-helix motifs known as EF-hands able to sense the Ca2+ fluctuations which are indispensable for activating a large group of Ca2+-binding proteins (Yanez et al., 2012). In CaNB, the first 2 EF-hands at the N-terminal lobe serve both as low affinity Ca2+-sensing motifs, while the other 2 EF-hands at the C-terminal lobe bind Ca2+ with high affinity with a constant of dissociation (Kd) in the nanomolar range (Kakalis et al., 1995). As suggested in different studies, the two pairs of motifs have different roles: the low affinity motifs have a regulatory role serving as sensors of the calcium fluctuations, whereas the high affinity motifs on the C-terminal lobe serve for stabilisation of the heterodimer (Stemmer & Klee, 1994; Klee et al., 1998).
The activation of CaN relies on a sequential structural rearrangement in which the first step is represented by the binding of two calcium ions to both low affinity EF-hands at the N-terminal lobe of CaNB. This event induces the opening of the CaM binding region that is then available for the recognition by CaM, which, in turn, promotes the displacement of the autoinhibitory domain from the catalytic site of CaNA (Stemmer & Klee, 1994; Yang & Klee, 2000). CaNB is indispensable for the catalytic activity of CaNA, and its genetic deletion has been widely used to knock-out CaN in different tissues and organs (Zeng et al., 2001; Chang et al., 2004; Heit et al., 2006; Parsons et al., 2007; Maillet et al., 2010).
Considering the biological relevance of CaN, it is easy to deduce that it can interact with a wide broad of proteins. In the years, many substrates, involved in a plethora of signal transduction pathways, were characterized, highlighting the key role of this enzyme in cellular homeostasis (Ingebritsen & Cohen, 1983; Goto et al., 1985; Pallen & Wang, 1985; Klee et al., 1988; Jain et al., 1993; Lieberman & Mody, 1994; Coghlan et al., 1995; Wang et al., 1999; Bhattacharyya et al., 2009). Among the CaN interactors, the most studied is the nuclear factor of activated T-cells (NFAT). The NFAT proteins are a family of transcriptional factors largely expressed in different cells of the immune system, where they were first identified (Shaw et al., 1988). Subsequently, it was found that NFAT is present in other cell types, including those of the nervous system (Ho et al., 1994). The NFAT family is formed by five members: NFATc1 (NFAT2, NFATc), NFATc2 (NFAT1, NFATp), NFATc3 (NFAT4, NFATx), NFATc4 (NFAT3) and NFAT5 (TonEBP: tonicity element binding protein or OREB: osmotic response element binding protein) (Im & Rao, 2004). The NFATc1-4 proteins are dephosphorylated at the N-terminal portion by activated CaN (Clipstone & Crabtree, 1992; Klee et al., 1998). This dephosphorylation induces a structural rearrangement of the proteins leading to the exposure of the nuclear localization sequences (NLSs) allowing the nuclear import of NFAT. The nature of the transcriptional modulation induced by NFAT could be either activating or repressive depending on the transcription factors which interact with NFAT in DNA binding, suggesting a context-dependent regulation. Indeed, NFAT activates the transcription when it cooperates with activator protein-1 (AP-1; an heterodimer formed by Fos and Jun), GATA proteins, MEF-2 and histone deacetylases (HDACs) (Blaeser et al., 2000; McKinsey et al., 2000; Avni et al., 2002; Hogan et al., 2003; Lee et al., 2003), but it also acts as a transcriptional repressor when the complex with AP-1 is disrupted (Macián et al., 2002).
The dephosphorylation of NFATs and their activation could be prevented by immunosuppressive drugs: cyclosporine A (CsA) and FK506 (tacrolimus). Both CsA and FK506 form complexes with the immunophilins cyclophilin A and FKBP12, respectively. These drug–immunophilin complexes then bind to and inhibit CaN (Griffith et al., 1995; Kissinger et al., 1995; Huai et al., 2002). Because both CsA and FK506 inhibit the phosphatase CaN activity directed to all the targets, their effect is not specific to NFAT (Martinez-Martinez & Redondo, 2004). For the selective inhibition of the signalling prompted by NFAT, specific synthetic peptides, able to compete with NFAT for CaN binding and to inhibit NFAT dephosphorylation, were developed (Aramburu et al., 1998; Garcia-Cozar et al., 1998). NFAT has two CaN binding sites: the PxIxIT motif at the N-terminal and the LxVP motif at the C-terminal region of the protein. The first characterized PxIxIT motif in NFATc2 was the PRIEIT peptide, whose overexpression prevents NFAT nuclear localization by competing with NFAT itself for the binding with CaN (Aramburu et al., 1998). Thereafter, a randomized PxIxIT motif library was generated leading to the identification of an optimized peptide called PVIVIT that has a 50-fold higher affinity for CaN than PRIEIT. Aramburu et colleagues demonstrated that the replacement of the native PRIEIT sequence with PVIVIT in NFAT induces the constitutive CaN-dependent dephosphorylation of NFAT and its permanent translocation inside the nucleus (Aramburu et al., 1999). When the PVIVIT peptide alone is overexpressed in the cell, it blocks specifically the NFAT-dependent pathway. In fact, it has become a powerful tool to inhibit NFAT-dependent processes both in vitro and in vivo(Furman et al., 2012; Furman et al., 2016). While the PxIxIT motif is considered to be the primary CaN-binding site for all members of the NFAT family, the LxVP motif seems to act as a regulator which modulates the strength of the CaN/NFAT-binding (Martínez-Martínez et al., 2006). Although the studies centred on the characterization of the synthetic peptides demonstrate the involvement of both PxIxIT and LxVP in NFAT binding to CaN, the functional interaction between these two sites remains to be fully understood.
Ca2+ signals and CaN activation
As suggested by the name, CaN is a Ca2+-dependent enzyme, and it is therefore important to understand the sources and spatio-temporal properties of Ca2+ signals implicated in its activation. The experiments dissecting the Ca2+ signals necessary for CaN-dependent gene transcription have mostly been performed in lymphocytes by either stimulating T-cell (or B-cell) receptor or by using inhibitors of the sarco-endoplasmic reticulum Ca2+ ATPase (SERCA) to induce [Ca2+]i elevation. A receptor mediated Ca2+ signal, which results from the inositol-1,4,5-trisphosphate (InsP3)-mediated Ca2+ release from the internal stores, has the shape of a transient rise which decays to the baseline after reaching a maximum of amplitude. For long-lasting Ca2+ signals, monitored as a monotonic plateau or Ca2+ oscillations, specific mechanisms have to be engaged,such as Ca2+-induced Ca2+ release (CICR) and/or store-operated Ca2+ entry(SOCE) (see below). Activation of the T cell receptor (TCR) may result in either a single Ca2+ transient or a transient followed by a long-lasting plateau or oscillations. Using the β-galactosidase reporter gene (lacZ) under the control of NFAT element of the interleukin-2 (IL-2) enhancer, Negulescu and colleagues demonstrated that CaN/NFAT-mediated gene expression depends on a long-lasting elevation of [Ca2+]i but not on a short Ca2+ transient (Negulescu et al., 1994). In this study it was elegantly demonstrated that T cells stimulated with immobilized anti-CD3 antibody exhibited a sustained Ca2+ signal that lasted for more than 25 min and promoted lacZ expression. Using the SERCA blocker thapsigargin (TG) to fully activate SOCE and clamping extracellular Ca2+ at different [Ca2+], the authors discovered that a sustained elevation of [Ca2+]i ranging between 0.2 and 1.6 μM for at least 10 min is necessary for CaN/NFAT to be activated (Negulescu et al., 1994). CaN activates several distinct signalling cascades, the most important of which are mediated by the transcription factors NFAT and NF-kB. An outstanding question is how the Ca2+ signals selectively discriminate between these two CaN targets. Dolmetsch and Lewis with their colleagues were the first to address this question. They found that, in B lymphocytes, the amplitude and the duration of Ca2+ signals differentially control the activation of NFAT versus NF-kB. NFAT was specifically activated by a low, sustained Ca2+ plateau, while NF-kB required a large Ca2+ transient (Dolmetsch et al., 1997). While Ca2+/CaN is the only way to activate NFAT, NF-kB-dependent gene transcription is the result of a multistep signalling cascades (see below), in which CaN can intervene at different steps. The refinement of the Ca2+ sensitivity of NFAT and NF-kB may be achieved by modulating the frequency and the amplitude of Ca2+ oscillations. Thus, low frequency oscillations activate preferentially NF-kB, while rapid oscillations activate both the transcription factors. Since Ca2+ oscillations have many more parameters to be modulated by specific mechanisms than the monotonic Ca2+ plateau (Dragoni et al., 2011; Smedler & Uhlén, 2014), they are widely used by different cell types, including astrocytes, in order to provide the ultimate specificity for Ca2+-regulated processes.
One of the important factors that, alongside the classical Ca2+ signalling toolkit, shapes the intracellular Ca2+ signals is represented by mitochondria, which are also denominated as the cellular “Ca2+ firewall” (Straub et al., 2000; Walsh et al., 2009). Mitochondria act as a high capacity and low affinity Ca2+ buffer which absorbs high quantities of Ca2+ ions generated locally in the proximity of Ca2+ channels of both the ER membrane and the plasma membrane. By their fast Ca2+ uptake mitochondria smooth the steep Ca2+ overshoot and prevent the channels from inactivating, thus generating the prolonged elevation of Ca2+ required for NFAT activation in T-cells (Hoth et al., 2000). Whether a similar mechanism is also used by astrocytes remains to be investigated.
In pathology, the maladaptive remodelling of the Ca2+ signalling machinery may lead to exaggerated and uncontrolled Ca2+ signals and to non-specific activation of Ca2+-dependent proteases which can transform physiologic CaN signalling to a pathologic one (Carafoli, 2004; Brini et al., 2013). As reported in a series of studies, CaN may be constitutively activated via cleavage by the Ca2+-dependent protease calpain (Tallant et al., 1988; Wang et al., 1989; Lakshmikuttyamma et al., 2004). This, in fact, occurs in a number of pathological conditions in which Ca2+-dependent proteases are activated (Wu et al., 2007), including ischemic myocardium (Lakshmikuttyamma et al., 2003; Burkard et al., 2005) and experimental glaucoma (Huang et al., 2010). In brain pathology, calpain cleavage of CaN has been reported in brain ischemia (Shioda et al., 2006), excitotoxic neuronal death (Wu et al., 2004), and in Alzheimer’s disease (Liu et al., 2005; Mohmmad Abdul et al., 2011). However, it is not clear from these reports whether the calpain-mediated activation of CaN occurs only in neurons or also in astrocytes. This latter hypothesis is supported by the notion that calpain is expressed in astrocytes (Perlmutter et al., 1990; Lee et al., 2000; König et al., 2003) and that both calpain and CaN inhibition protect astroglial cells form Ca2+-reperfusion-induced apoptotic death (Takuma et al., 1999; Takuma et al., 2004).
Over the past decade, different fluorescence CaN reporters have been developed (Newman & Zhang, 2008; Lodygin et al., 2013; Mehta et al., 2014), none of which has, however, been tested in astrocytes. Using fluorescence resonance energy transfer (FRET)-based CaN sensors directed to different sub-cellular locations Zhang and colleagues have demonstrated the crucial role played by CaN distribution in CaN activation (Mehta et al., 2014).
Astroglial Ca2+signalling and Ca2+ routs for CaN activation
Astrocytes are the major cellular type in the brain outnumbering neurons by 2-8 folds(Verkhratsky, 2007).They perform fundamental housekeeping functions, including the maintenance of structural, metabolic and ionic homeostasis, and constitute an essential component of the neuro-vascular unit (Verkhratsky, 2007). Astrocytes have an extremely complex sponge-like morphology with a huge number of very fine processes (Bushong et al., 2002; Nedergaard et al., 2003). The unprecedented complexity of protoplasmic astrocytes does not only involve their morphological aspects (Bushong et al., 2002; Nedergaard et al., 2003), but it is becoming clear that the Ca2+ signalling architecture in these cells is extremely complex as well (Volterra et al., 2014; Shigetomi et al., 2016). Astrocytic processes ensheath synaptic structures forming the so called tripartite synapse – a structure composed of pre- and postsynaptic terminals and of astroglial processes, which insulate the synapse, providing the micro-environment for optimal synaptic functioning (Araque et al., 1999; Verkhratsky & Nedergaard, 2014). The tripartite structure provides a tool for communication between neurons and astrocytes. It has been hypothesized that astrocytes can play an active role in bidirectional astrocyte-neuron signalling by modulating the availability of neurotransmitters, thereby participating in information processing (De Pittà et al., 2016). The gliotransmission and the role of astroglial Ca2+ signals in modulation of synaptic transmission is a subject of intensive debates (Kimelberg, 2007; Agulhon et al., 2008; Fiacco et al., 2009; Pereira & Furlan, 2010; Agulhon et al., 2012; Benneyworth et al., 2012; Nedergaard & Verkhratsky, 2012; Ota et al., 2013; Araque et al., 2014;Fujita et al., 2014; Perea et al., 2014; Sloan & Barres, 2014; Volterra et al., 2014;Oliveira et al., 2015). Elevations of [Ca2+]i in astrocytes, which may be associated with CaN activation, are frequently found in acute and chronic neurodegenerative pathologies.In this section, we discuss the mechanisms of generation of Ca2+ signals in astrocytes in the context of their possible role in CaN activation.
Calcium is a universal second messenger which regulates a vast spectrum of cellular processes (Fedrizzi et al., 2008; Brini et al., 2013). The specificity of the signal, which is also defined as information encoding, is assured by the organ/cell-specific composition of the so called Ca2+signalling toolkit (Berridge et al., 2003; Verkhratsky et al., 2012a) which generates a stimulus-specific rise of [Ca2+]i precisely defined in time and space. The changes of [Ca2+]i in astrocytes have been associated with virtually all physiological functions ranging from release of gliotransmitters (Parpura & Haydon, 2000) to astroglial plasticity (Pirttimaki & Parri, 2013) and responses to sensory stimulation (Lind et al., 2013).
Being electrically non-excitable cells, in the sense that they do not trigger a voltage-dependent action potential, which is a characteristic of neurons, astrocytes respond to extracellular stimuli by employing mainly metabotropic Ca2+signalling and the membrane of the endoplasmic reticulum (ER) as an excitable media (Verkhratsky et al., 2012a). Astrocytes express a variety of metabotropic G-protein-coupled receptors (GPCRs) (Verkhratsky, 2007), of which, perhaps, the best studied are the group I metabotropic glutamate receptors (mGluRs) and the purinergic metabotropic receptors of the P2Y family. The binding of a ligand (e.g. glutamate or ATP) to a receptor triggers a signalling cascade which initiates with GTP-dependent dissociation of the trimeric G-protein. The αq/11 subunit of the G-protein binds to and activates PLCb, which in turn, hydrolyses phosphatidylinositol 4,5-bisphosphate (PIP2) in the plasma membrane, generating two second messengers, diacylglycerol (DAG) and InsP3. While DAG is involved in protein kinase C (PKC) signalling, InsP3, a soluble and diffusible second messenger, binds to and activates the InsP3 receptors (InsP3Rs) in the membrane of the ER, inducing release of Ca2+ from the ER lumen into the cytosol. Of the three known InsP3R isotypes, type 2 is considered to be the principal InsP3R in astrocytes in intact tissue(Holtzclaw et al., 2002; Petravicz et al., 2008), although presence and activity of InsP3R1 in cultured astrocytes has also been documented (Grolla et al., 2013b; Grolla et al., 2013a).
The InsP3-induced increase of [Ca2+]i, measured as a local Ca2+ transient, is the initial event which may be terminated without propagation (Verkhratsky et al., 2012a), but could be amplified within the same cell. Furthermore, the increase of [Ca2+]i may also propagate to the neighbouring astrocytes, generating inter-cellular Ca2+ waves (Charles, 2005; Scemes & Giaume, 2006), which are mainly mediated by InsP3 diffusion through gap junctions (GJ), alongside with the paracrine action of ATP (Cornell-Bell et al., 1990; Dani et al., 1992).
The amplification and propagation of Ca2+ signals within the astrocyte occur mainly via two mechanisms: i) the CICR and ii) the SOCE. Ca2+ ions, massively released from the ER through InsP3Rs, generate a local domain of high [Ca2+], the so called “hot spot” or “high Ca2+ microdomain” (Rizzuto et al., 1999; Parekh, 2008; Contreras et al., 2010). Diffusion of Ca2+ ions through the cytosol is severely restricted (Santella et al., 2003) due to potent buffering mechanisms (cytosolic Ca2+ buffers, Ca2+ uptake by juxtaposed mitochondria, Ca2+ transporters). However, Ca2+ ions are able to act at the neighbouring InsP3Rs, sensitizing them to a lower [InsP3], generating a Ca2+ wave, which propagates along the ER membrane (Verkhratsky & Kettenmann, 1996; Verkhratsky, 2007).
Another additional important mechanism to achieve a long-lasting Ca2+ elevation in the cytosol of astrocytes is SOCE, also known as capacitative Ca2+ entry (CCE) (Verkhratsky & Parpura, 2014). SOCE is triggered when Ca2+ is released from the ER after activation of InsP3Rs and the ER [Ca2+] drops down (Putney, 2007; Moccia et al., 2015). Stromal interacting protein 1 (Stim 1), an integral protein of the ER membrane, senses the decrease of intra-ER [Ca2+] and transmits the signal to a channel on the plasma membrane called Orai, of which three isoforms exist (Orai 1, 2, 3) (Putney, 2007; Moccia et al., 2015). Although it is postulated that the activation of Orai is essential for SOCE, another group of cation-permeable channels, the so called Transient Receptor Potential(TRP) channels may be involved in CCE(Salido et al., 2009; Cheng et al., 2013). In astrocytes, SOCE generates a long-lasting (minutes) Ca2+ elevation which is recorded as a plateau which follows the peak of the InsP3R-mediated Ca2+ transient (Ronco et al., 2014; Verkhratsky & Parpura, 2014). SOCE is a common feature present in all types of astrocytes (Tuschick et al., 1997; Pivneva et al., 2008; Verkhratsky & Parpura, 2014).Although all the components of the SOCE molecular machinery, including STIM, Orai and TRP channels (Pizzo et al., 2001; Grimaldi et al., 2003; Golovina, 2005; Barajas et al., 2008; Malarkey et al., 2008; Ronco et al., 2014; Verkhratsky et al., 2014), are present in astrocytes, experiments suggest that astroglial cells employ mainly TRP channels to generated SOCE (Verkhratsky & Parpura, 2014) and to coordinate ion signalling and astroglial excitability (Verkhratsky et al., 2014). Intriguingly, SOCE is the main mechanism for CaN activation in peripheral immune cells and lymphocytes (Gwack et al., 2007; Oh-hora & Rao, 2008).
Another form of Ca2+ signals which is common for both the individual astrocytes and the astroglial networks are Ca2+ oscillations (Zonta & Carmignoto, 2002; Koizumi, 2010). The mechanisms of CICR and SOCE have been generally implicated in setting up the oscillatory Ca2+ dynamics (Carafoli et al., 2001; Uhlen & Fritz, 2010; Dupont et al., 2011; Parekh, 2011). Specifically in astrocytes, TRP channels are involved in glutamate-induced Ca2+ oscillations (Pizzo et al., 2001). Ca2+-dependent release of ATP, instead, is implicated in synchronized Ca2+ oscillations in astroglial networks (Koizumi, 2010).
Ca2+ entry from the extracellular medium via ligand-gated and voltage-gated Ca2+channels may generate Ca2+ signals in astrocytes (James & Butt, 2002; Verkhratsky, 2007; Palygin et al., 2010; Verkhratsky et al., 2012b). The expression of Ca2+ ion channels in astrocytes is subtype- and region-specific. It has been suggested that most types of astrocytes express L-type and T-type Ca2+ currents (Verkhratsky &Steinhauser, 2000). Whilethe expression of different types of Ca2+channels was found in a population of immature mouse hippocampal astrocytes, apparent absence of Ca2+ currents was found in adult rat hippocampal and visual cortex astrocytes (reviewed in Verkhratsky& Steinhauser, 2000). Ca2+ permeable AMPA receptors/channels have been found in oligodendrocytes and NG2 glia (Lin& Bergles, 2002). Astrocytes in cortex and spinal cord express NMDA receptors (Verkhratsky & Kirchhoff, 2007).Stimulation of nicotinic acetylcholine receptors (nAChRs) either with nicotine or with acetylcholine has been shown to elevate astroglial [Ca2+]i (Teaktong et al., 2003; Oikawa et al., 2005). A series of reports suggest that astroglial nAchRs can be target of Aβ in AD (reviewed in Sadigh-Eteghad et al., 2016). Culturedastrocytes express different types of purinergic P2X receptors (P2X1-5,7). In intact tissue, P2X receptors have been found in astrocytes of many brain regions including cortex, hippocampus, cerebellum and brainstem (reviewed in Illes et al., 2012). Ionotropic receptors in astrocytes are mainly localized in fine processes adjacent to the post-synaptic terminals and it is not known whether they participate in the propagation of Ca2+ signals through the astrocyte and in CaN activation. In addition, Ca2+ permeability of ligand-gated channels in astrocytes is relatively low with respect to Na+(Kirischuk et al., 2012), which makes them more suitable for controlling Na+ homeostasis.
Another route for generating intracellular Ca2+ signals, which appears to be specific for astrocytes, is represented by the Na+/Ca2+ exchanger (NCX). NCX is an antiporter that exploits the energy of the electrochemical gradient of Na+ to extrude Ca2+ out from the cell. To expel one Ca2+ ion, NCX allows three Na+ ions to enter (Annunziato et al., 2004; DiPolo & Beauge, 2006). In the majority of cellular types NCX constitutes one of the most important mechanisms of extruding Ca2+ from the cells representing low affinity and high capacity Ca2+ transport system. It is, therefore, different from the plasma membrane Ca2+ ATPase pump (PMCA) which has high Ca2+ affinity but relatively low transport capacity. In astrocytes, NCX functions in the reverse mode,transporting Na+ out and allowing Ca2+ to flow into the cell (Takuma et al., 1994; Reyes et al., 2012). This occurs due to low reversal potential for NCX (~ -87 mV), which is just slightly more negative than the resting membrane potential (-80 to -85 mV). Increases of [Na+]i (e.g., after the activation of Na+-permeableionotropic receptors, voltage-gated Na+channels andNa+-dependent glutamate transporters)reduce the electrochemical Na+ gradient, therebyallowing Ca2+ to enter the cell (Annunziato et al., 2004; DiPolo & Beauge, 2006; Verkhratsky et al., 2012a). However, at present it is not known whether NCX-mediated Ca2+ signals contribute to CaN activation in astrocytes.
Taken together, astrocytes are endowed with versatile and stimulus-specific mechanisms to encode the signalling information within Ca2+ signals (Figure 1). In the following sections we will discuss how these signals are decoded by CaN and by its downstream targets to achieve specific transcriptional and functional outputs.
Figure 1. Astroglial Ca2+ routes for CaN activation The principal mechanism of CaN activation in astrocytes is represented by the activation of the seven transmembrane domains G-protein coupled receptors (GPCR), which generates the second messenger InsP3 that in turn activates the InsP3Rs on the ER membrane (depicted as thin green arrows). Once activated, the InsP3Rs induce the store-operated Ca2+ entry, which involves the interaction between the STIMs molecules onto the ER and the Orai channels located into the plasma membrane. That recruits the transient receptor potential (TRP) channels to induce a massive Ca2+ entry (depicted as bold green arrow). Ca2+-induced Ca2+ release (depicted as light-green thin arrows) may also be involved in the amplification of Ca2+ signal necessary for CaN activation, whereas the roles of mitochondrial Ca2+ uptake (Mit), ligand gated Ca2+ channels (LGCC) and sodium/calcium exchanger (NCX) is not clear. Note that IP3R1 has been studied in cultured astrocytes while in intact tissue IP3R2 is the principal isoform
Calcineurin signalling in astrocytes
Ca2+, CaN and NFAT
The term “calcineurin” was coined by Claude Klee and co-workers in 1979 “on the basis of its Ca2+-binding properties and its specificity for the nervous system” (Klee et al., 1979). In later years, CaN was found to be expressed in many (if not all) cellular types, including astrocytes and microglia. Curiously, initial examination of the brain using anti-CaN antibody failed to detect CaN in astrocytes (Goto et al., 1986b), although experiments using pharmacological inhibitors of CaN suggested its presence (Randriamampita & Tsien, 1995; Hosoi et al., 1997; Matsuda et al., 1998). Thereafter, CaN protein was found in cultured astrocytes and in rodent brain in situ(Hashimoto et al., 1998), and presence of both the catalytic CaNA and the regulatory CaNB subunits was confirmed (Vinadé et al., 1997; Matsuda et al., 1998). As determined by microarray and RNA-sequencing, at mRNA level, astrocytes express high levels of the two principal isoforms of the catalytic subunit, CaNAα and CaNAβ, and the CaNB1 isoform of the regulatory subunit, while CaNAγ and CaNB2 mRNA are present at much lower levels (Cahoy et al., 2008; Orre et al., 2014; Zhang et al., 2014). Both CaNAα and CaNAβ proteins are expressed in cultured astrocytes (Canellada et al., 2008; Lim et al., 2013).
As it was mentioned above, NFAT is the direct and, at the same time, the most studied target of CaN. Numerous publications reported the expression of NFAT in astrocytes and astroglial cell lines (Jones et al., 2003; Canellada et al., 2008). At the mRNA level, both developing and adult astrocytes express all four NFAT isoforms although their overall amount is lower than that of CaN (Cahoy et al., 2008; Orre et al., 2014; Zhang et al., 2014). At the protein level, NFATc3 has been detected in most studies (Jones et al., 2003; Filosa et al., 2007; Canellada et al., 2008; Serrano-Pérez et al., 2011; Neria et al., 2013; Yan et al., 2014; Furman et al., 2016),while the expression of NFATc1 (Pérez-Ortiz et al., 2008) and NFATc2 (Furman et al., 2016) has also been reported.
CaN and, consequently, NFAT in astrocytes may be activated by a variety of stimuli including neuro(glio)transmitters (glutamate and ATP) (Jones et al., 2003; Filosa et al., 2007; Canellada et al., 2008; Pérez-Ortiz et al., 2008), inflammatory agents (IL-1β) (Sama et al., 2008), experimental manipulations using a Ca2+ ionophore alone (Jones et al., 2003; Pérez-Ortiz et al., 2008) or in combination with phorbol myristate acetate (PMA), an activator of PKC and a wide range of its downstream kinases (Neria et al., 2013).
Treatment of cultured astrocytes with β-amyloid (Aβ) peptide, which is thought to be the main cause of AD (Selkoe & Hardy, 2016), induced the rapid translocation of NFAT into the nucleus (Abdul et al., 2009). In the human astrocytoma U373MG cell line, Aβ-induced activation of the CaN/NFATc3 axis has been shownto elicit the expression of beta-site amyloid precursor protein (APP) cleaving enzyme 1 (BACE1) (Jin et al., 2012), which is involved in the over-production of toxic Aβ(1-42) (Aβ42). In this context, it has been demonstrated that rat hippocampal astrocytes in culture express APP itself and all the enzymatic machinery to process APP and to generate and release β-amyloid peptide in a cell autonomous manner (Grolla et al., 2013b).
Simulation of acute brain pathology by injection of kainic acid and mechanical lesion triggers NFATc3 over-expression in vivo in a subset of reactive astrocytes (Serrano-Pérez et al., 2011; Neria et al., 2013). In another in vivo model, the ischemia-reperfusion, Rcan1-4 was induced in GFAP-positive astrocytes in the site of lesion (Sobrado et al., 2012). Regulation of Rcan1-4 and COX2 gene expression in astrocytes has been shown to be controlled by CaN/NFATc3 in cultured(Canellada et al., 2008) and in situ (Sobrado et al., 2012) astrocytes.
Using traumatic brain injury (TBI) with controlled cortical impact (CCI), Furman et al (Furman et al., 2016) observed over-expression of NFATc3 and its increased DNA binding in hippocampal lysates at 1 week after CCI TBI. Immunohistochemical analysis confirmed that the over-expressed NFATc3 was localized in astrocytes. Furthermore, adeno-associated virus (AAV)-mediated astrocyte-specific expression of VIVIT, a peptide which inhibits NFAT-CaN interaction thereby precluding NFAT activation (Aramburu et al., 1999) (see above), significantly reduced TBI-induced nuclear translocation of NFATc3 (Furman et al., 2016). However, using the same CCI TBI protocol, Yan et al (Yan et al., 2014) found down-regulation of NFATc3 at all the time-points (from 6 hours to 4 weeks) after CCI TBI in both cytosolic and nuclear hippocampal fractions.Immunohistochemical analysis showed astroglial localization of NFATc3 and it is concluded that the astroglial NFATc3 signalling is down-regulated after CCI TBI (Yan et al., 2014). The reasons for such a discrepancy are not clear. These are the first reports that use the CCI TBI protocol of brain injury to monitor NFAT activation and further experiments are warranted to confirm the CCI TBI-induced NFATc3 activation in astrocytes.
Among the transcriptional targets of CaN/NFAT signalling in astrocytes there are Rcan1-4 (Canellada et al., 2008; Serrano-Pérez et al., 2011; Neria et al., 2013), COX2 (Blanco et al., 2008; Canellada et al., 2008; Serrano-Pérez et al., 2011), TNFα, GM-CSF, S100β, Vimentin (Sama et al., 2008), IL-6 (Serrano-Pérez et al., 2011), BACE1 (Jin et al., 2012), and MMP3 (Neria et al., 2013). All these genes were up-regulated after activation of NFAT. Instead, GLT-1 (EAAT2) was down-regulated when NFAT was activated by either IL-1β or β-amyloid oligomers (Sama et al., 2008; Abdul et al., 2009). Ca2+ entry appears to be essential for the above effects since Ca2+ channel blockers (Canellada et al., 2008; Sama et al., 2008) or a Ca2+ chelator (Jin et al., 2012) abolished NFAT activation in cultured astrocytes. In other cases, to achieve CaN/NFAT activation, Ca2+ entry was forced directly by an ionophore (Canellada et al., 2008; Neria et al., 2013) or indirectly by kainic acid lesion (Serrano-Pérez et al., 2011) (See Table 1).
Table 1. CaN regulation of NFAT/NF-kB-mediated signaling in astrocytes.
|
Reference |
Stimulus (in vitro or in vivo) |
Exp. time |
Mediator (Inhibitor) |
NFAT or NF-kB |
CaN Inhibitor |
Target genes or effects |
Up or Down |
|
Norris et al., 2005 microarray |
Overexpression ΔCaNA |
48 h |
CaN |
NFAT |
|
Morphogenesis;Ion homeost.; Immune resp.;Cell adhesion. |
UP |
|
Biosynthesis;TCA cycle; Intracell. Sign. cascades |
DOWN |
||||||
|
Blanco et al., 2008 |
TAT |
16 h |
|
NFAT |
|
COX2 |
UP |
|
Canellada et al., 2008 |
PMA+Iono |
4 h |
Ca2+, PKC |
NFAT |
|
Rcan1-4, COX2 |
UP |
|
BayK 8644 |
4 h |
VGCC (Nifedipine, Verapamil) |
NFAT |
|
Rcan1-4, COX2 |
UP |
|
|
Sama et al., 2008 |
Il-1β |
3 h |
VGCC (Nifedipine) |
NFAT |
VIVIT |
TNFa, GM-CSF, S100B, Vimentin |
UP |
|
GLT-1 (EAAT2) |
DOWN |
||||||
|
Abdul et al., 2009 |
Aβ42 (65 nM) |
24 |
|
NFAT |
VIVIT |
GLT-1 (EAAT2) |
DOWN |
|
Serrano-Perez et al., 2011 |
Kainic acid injury |
6-24 h |
|
NFAT |
VIVIT |
Rcan1-4, COX2, IL-6 |
UP |
|
Jin et al., 2012 |
Aβ42 (1 uM), Aβ25-35 (20 uM) |
24 h |
Ca2+(BAPTA) |
NFATc3 |
CsA |
BACE1 |
UP |
|
Neria et al., 2013 |
Overexpression NFATc3; PMA+Iono, but not LPS |
6 h |
|
NFATc3 |
CsA |
MMP3, Rcan1-4 |
UP |
|
Takuma et al., 1999 |
Ca2+ reperfusion injury |
|
CaN |
NF-kB |
PDTC FK506 |
Apoptosis |
|
|
Alvarez et al., 2008 |
HIV-2 |
16 h |
CXCR4 |
NF-kB |
Overexpr IkBα |
COX2 |
UP |
|
Blanco et al., 2010 |
Aβ42 100 nM (0.5 ug.ml) |
16 h |
|
NF-kB |
PDTC |
COX2 |
UP |
|
Lim et al., 2013 |
Aβ42 100 nM |
24 h |
Ca2+-CaN |
NF-kB |
JSH-23, CAPE |
mGluR5, InsP3R2 |
UP |
A microarray approach was undertaken by Norris et al (Norris et al., 2005) to find CaN-dependent genes in mixed neuron-astrocyte cultures in which astrocytes were transduced with adenovirus (Ad) encoding a truncated ~ 45 kD constitutively active CaN fragment (ΔCaN). Of 3403 genes analyzed across the three conditions (non-transduced, Ad-LacZ and Ad-ΔCaN), 719 were differentially expressed between the Ad-LacZ and the Ad-ΔCaN groups. The more stringent group of 298 genes was selected in the Ad-ΔCaN set which differs from both non-transduced and Ad-LacZ to eliminate probable impact of viral infection on gene expression (Norris et al., 2005). Among the most overrepresented gene ontology (GO) terms in this dataset there were: morphogenesis, ion homeostasis, immune response and cell adhesion for up-regulated genes; while the GO terms lipid biosynthesis, tricarboxylic acid cycle and intracellular signalling cascade were overrepresented in down-regulated genes (Norris et al., 2005). Somewhat surprising is that among the aforementioned CaN/NFAT transcriptional targets, only two genes, namely the renowned astroglial markers S100β and vimentin, were differentially regulated by the overexpression of ΔCaN. This may be tentatively explained by the differential recruitment of CaN to stimulus-specific pathways. For example, PMA plus ionophore treatment, which produce robust Ca2+ signals and phosphorylation, induces Rcan1-4 and COX2, but not InsP3R1 expression (Canellada et al., 2008). Instead, Aβ42-induced and CaN-mediated up-regulation of InsP3R1 in astrocytes (Grolla et al., 2013b; Grolla et al., 2013a) require low and sustained elevation of [Ca2+]i.
Ca2+, CaN and NF-kB
NF-kB is a family of closely related transcription factors with an established role in innate immunity and inflammation (Newton & Dixit, 2012; Hoesel & Schmid, 2013). The family consists of hetero- and homodimers composed of five monomers, RelA (p65) (encoded by gene Rela), RelB (Relb), cRel (Rel), p50 and p52. The most important heterodimers are RelA:p50, cRel:p50 and RelB:p52 (Shih et al., 2011). In addition to transcriptional regulation of monomer’s availability, p50 and p52 are also regulated by processing of the precursor proteins p105 (encoded by Nfkb1) and p100 (encoded by Nfkb2), respectively.
The signalling cascade responsible for NF-kB activation may be triggered by a number of stimuli including pro-inflammatory agents like TNFα, IL-1β and LPS, and by stimulation of T-cell or B-cell receptors (TCR and BCR) (Paul & Schaefer, 2013). The molecular cascades leading to NF-kB activation are complex and cell-specific, yet somewhat non-linear. The canonical NF-kB pathway, in response to TCR activation, converges on a trimeric molecular complex composed of CARMA1, MALT1 and Bcl10 proteins (CBM complex) (Rosebeck et al., 2011). The formation of CBM complex, through intermediate passages, results in phosphorylation of IKKβ, which phosphorylates the inhibitor of kBα (IkBα), whose consequent ubiquitination and proteasomal degradation enables nuclear translocation of a p65/p50 heterodimer, which interacts with partner transcription factors to drive the expression of numerous genes. It has long known that, under experimental conditions, the addition of a Ca2+ ionophore significantly facilitates NF-kB activation, thus establishing a role for Ca2+ in NF-kB activation. In fact, it is a general laboratory practice to induce NF-kB activation in a cell culture by stimulation with PMA (which stimulates PKC and downstream cascades) in combination with the Ca2+ ionophores A23187 or ionomycin. Ca2+-dependent steps in TCR-triggered NF-kB activation include initial stabilization of CMB complex by CaMKII-mediated phosphorylation of CARMA1 and Bcl10 with consequent dissociation and IKKβ phosphorylation (Paul & Schaefer, 2013). CaN-mediated dephosphorylation of Bcl10 is required for CBM complex stability in canonical signalling from TCR to NF-kB (Frischbutter et al., 2011; Palkowitsch et al., 2011).
A number of works reported that in astrocytes NF-kB activation by LPS, IL-1β or TNFα: i) requires Ca2+/CaM-dependent enzymes; and that ii) this requirement may be context-specific. For examples, Liu and colleagues (Liu et al., 2000) reported the involvement of Ca2+ signalling in IL-1β-induced NF-kB-dependent transcription of TNFα and IL-6 in cultured human fetal astrocytes, whereas Schwaninger and colleagues (Schwaninger et al., 1999) found that Ca2+/CaM dependent protein kinases are essential for bradykinin-induced NF-kB activation and IL-6 induction in murine astrocytes. As reported by (Nath et al., 1999), NF-kB-dependent induction of IL-6 but not IL-1β production was promoted by HIV-1 Tat protein independently on extracellular Ca2+, which however does not rule out the requirement for Ca2+ released from internal stores. In fact, El-Hage et al showed that NF-kB-mediated production of MCP-1 and IL-6 by HIV Tat is highly Ca2+-dependent and that opiates potentiate Tat-induced NF-kB activation via a Ca2+-dependent pathway (El-Hage et al., 2008).
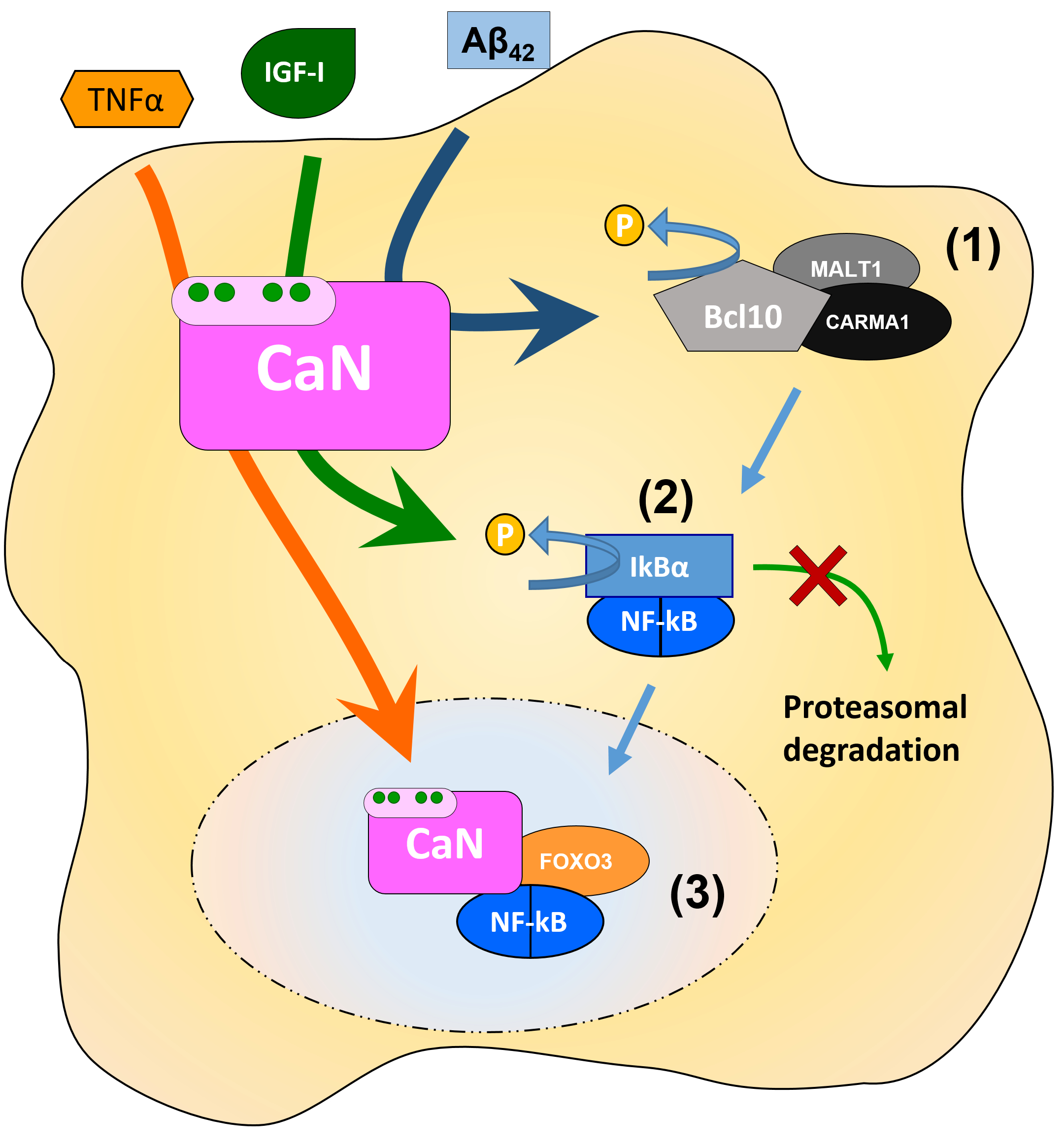
Although the entire cascade responsible for NF-kB activation in astrocytes is not yet fully identified, available literature suggests that CaN can intervene in NF-kB signalling at several points (Figure 2): i) CaN dephosphorylates Bcl10, which positively regulated NF-kB signalling in hippocampal cultured astrocytes(Lim et al., 2013); ii) CaN dephosphorylates IkBα, thus precluding its degradation which inhibits NF-kB nuclear translocation in culture(Pons & Torres-Aleman, 2000); iii) CaN, in complex with NF-kB and FOXO3, is required for TNFα-induced NF-kB nuclear translocation and activation of transcriptionin vitro and in vivo(Fernandez et al., 2012). In the first case, the co-immunoprecipitation assay suggests constitutive interaction of both CaNA isoforms, α and β, with Bcl10 in cultures of hippocampal astrocytes. Importantly, an amount of co-precipitated Bcl10 increased upon treatment with 100 nM Aβ42 oligomers, while the phosphorylation status of Bcl10 decreased (Lim et al., 2013). At the same time, Aβ42 induced the increase of [Ca2+]i followed by CaN/NF-kB-dependent up-regulation of key components of the astroglial Ca2+signalling toolkit, such as mGluR5 and InsP3R2 (Lim et al., 2013; Ronco et al., 2014), suggesting a role for CaN/NF-kB axis in Aβ-induced deregulation of astroglial Ca2+signalling in AD (Lim et al., 2014; Lim et al., 2016). Interestingly, in hippocampal astroglial cultures, pro-inflammatory agents such as TNFα, IL-1β and LPS, induced NF-kB-dependent down-regulation of mGluR5, InsP3R1 and InsP3R2 mRNA as well as of DHPG-elicited Ca2+ signals (Ronco et al., 2014). However, in these case TNFα, IL-1β or LPS-induced degradation of IkBα was CaN-independent (Ronco et al., 2014) (Table 2). mGluR5 has previously been shown to be down-regulated by TNFα, IL-1β and LPS in astrocytes in culture(Aronica et al., 2005; Tilleux et al., 2007; Berger et al., 2012).
Figure 2. Schematic illustration of specific points in the NF-kB astroglial signalling cascade in which CaN can intervene (1) CaN dephosphorylates Bcl10, which potentiates NF-kB signalling in hippocampal cultured astrocytes (Lim et al., 2013); (2) CaN dephosphorylates IkBα, thus precluding its degradation which results in the inhibition of NF-kB nuclear translocation (Pons & Torres-Aleman, 2000); (3) CaN, in complex with NF-kB and FOXO3, is required for TNFα-induced NF-kB nuclear translocation and activation of transcription both in vitro and in vivo(Fernandez et al., 2012)
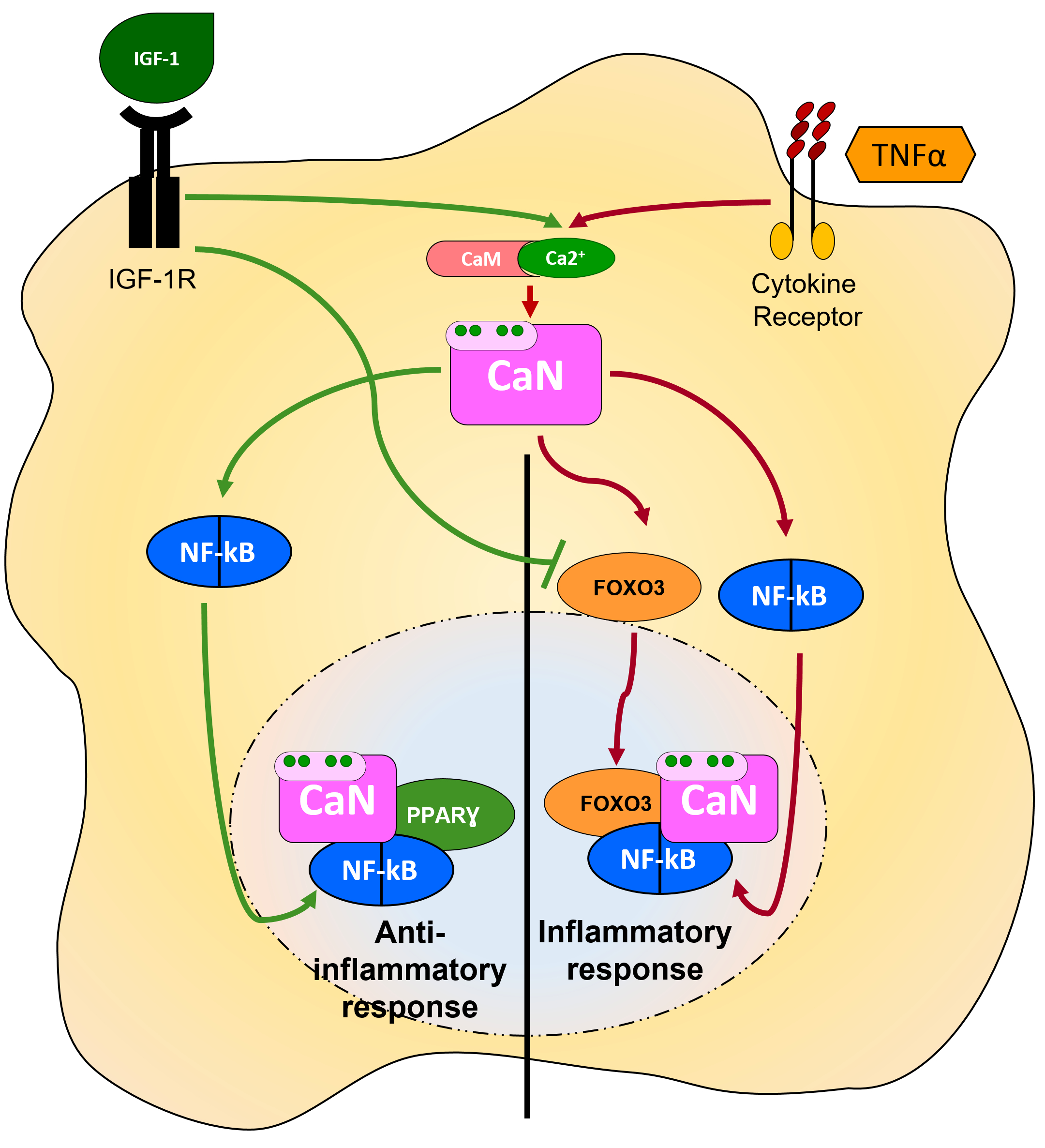
A dual nature of CaN as activator or inhibitor of NF-kB/NFAT signalling in astrocytes has been accurately dissected in a series of reports from Torres-Aleman’s group (Pons & Torres-Aleman, 2000; Fernandez et al., 2007; Fernandez et al., 2012) (Figure 3). Such an ambivalent functional outcome of CaN activation is set up by the equilibrium between pro- or anti-inflammatory environments(Fernandez et al., 2012; Furman & Norris, 2014). While the canonical pro-inflammatory pathway, induced by TNFα or LPS, requires CaN to mediate the NF-kB/NFAT-dependent induction of iNOS and COX2, this effect is counteracted by the anti-inflammatory mediator IGF-I, which recruits CaN to inhibit NF-kB/NFAT mediated inflammation (Fernandez et al., 2007). In astrocytes, IGF-I-activated CaN dephosphorylates IkBα at serine 32, thus precluding its degradation and NF-kB activation. Interestingly, IGF-I-induced CaN activation in astrocytes requires neither PI3-kinase or Akt (Okubo et al., 1998; Pons & Torres-Aleman, 2000), nor p38 or PLCγ, although elevation of [Ca2+]i is essential for the effect since chelating intracellular Ca2+ with BAPTA-AM has the same inhibitory effect on IGF-I-induced CaN activation as inhibition of CaN itself with CsA (Pons & Torres-Aleman, 2000). The exact mechanism by which IGF-I generates intracellular signals in astrocytes to activate CaN is, however, yet to be elucidated. When focusing on the IGF-I-induced inhibition of NF-kB, a specific role of FOXO3 and PPARγ has been recently investigated. TNFα stabilizes the interaction of CaNA with FOXO3 and NF-kB generating a complex, which drives the transcriptional activity of NF-kB (Fernandez et al., 2012). Interestingly, both CaN and FOXO3 are required for TNFα-induced NF-kB activation and induction of inflammation-associated genes like COX2 (Fernandez et al., 2012). In this context, TNFα-induced CaN activation does not affect degradation of IkBα (Ronco et al., 2014). To counteract TNFα-induced NF-kB activation, in addition to the dephosphorylation of IkBα by CaN, IGF-I recruits PPARγ to displace FOXO3 from the CaNA/NF-kB complex (Fernandez et al., 2007). In the complex CaNA/NF-kB/PPARγ, PPARγ is sumoilated which results in trans-repression of NF-kB transcription (Glass & Ogawa, 2006; Fernandez et al., 2007). In according with data on cultured astrocytes, the interaction of CaN with NF-kB and FOXO3 occurs in vivo in the brain of the AD model APP/PS1 mice. Moreover, the complex is disrupted by IGF-I or by over-expression of ΔCaNA (Fernandez et al., 2012). The functional outcomes of the ΔCaNA-induced NF-kB inhibition are the resolution of neuroinflammation and the alleviation of AD pathology. Recently, Fernandez and colleagues developed a series of decoy compounds which disrupted the interaction of CaN with FOXO3 in astrocytes thereby causinginhibition of NF-kB activation, resolution of inflammation and protection against Aβ-induced neurotoxicity in APP/PS1 mouse AD model (Fernandez et al., 2016).
It should be noted that most of the work on the characterization of CaN signalling in the brain has been done in cultured astrocytes or in whole brain tissues in which neurons are also present. Highlighting that the results obtained in cultures provide a fundamental information on interactions between proteins making up signalling cascades, one should be aware that the expression of these signalling components may differ from that of intact astrocytes in the brain. As an example, it has been demonstrated that the procedure of preparation of acute hippocampal brain slices per se induces reactive gliosis (Takanoet al., 2014).
Figure 3. Activation vs inhibition of NF-kB-mediated signalling: role of CaN interacting proteins Upon stimulation with TNFα, CaN recruits and dephosphorylates FOXO3 to interact with and activate NF-kB, which promotes transcription of inflammatory genes. When co-stimulated with IGF-I, CaN recruits PPARγ to displace FOXO3 from the CaN/FOXO3/NF-kB complex generating the CaN/PPARγ/NF-kB complex, thus precluding NF-kB transcriptional activity (Fernandez et al., 2012)
Can astroglial CaN be activated during physiological brain activity?
CaN signalling in astrocytes has been principally described in pathological or pathology-related experimental conditions in which CaN mediates a spectrum of signalling and functional outcomes. Conversely, it is still unclearwhether and how CaN signalling play any role in astroglial physiology. CaN is a renowned Ca2+/calmodulin-dependent master-regulator of cellular remodelling. Therefore, it is plausible to speculate that CaN could participate in structural and functional plastic changes, known also as astroglial plasticity (Shao & McCarthy, 1994; Theodosis & Poulain, 1999; Theodosis et al., 2008; Pirttimaki & Parri, 2013; Cheung et al., 2015), to which astrocytes undergo during neuronal activity. The hint that this could be the case can be taken from the landmark contribution by Filosa and colleagues reporting the CaN-dependent nuclear translocation of NFATc3, in both astrocytes and pericytes in cortical brain slices from ten days old rats,in response to electrical field stimulation (EFS) (Filosa et al., 2007). This effect was reproduced by treating the slices with the agonist of mGluR5 metabotropic glutamate receptors,t-ACPD. Interestingly, NFATc3 translocation in pericytes required functional astrocytes (Filosa et al., 2007). This is the first work reporting the activity-dependent activation of CaN in non-neuronal cells including astrocytes. However, the translation of these results to the activity-dependent CaN activation in adult healthy brain astrocytes is not straightforward. The expression of theCa2+-linked glutamate receptor mGluR5 is developmentally down-regulated and its transcript is not detectable after 3 weeks in mouse cortical and hippocampal astrocytes and in adult human astrocytes (Sun et al., 2013). This finding is supported by the failure of specific agonists (t-ACPD, DHPG) to elicit detectable Ca2+ responses in adult mouse astrocytes (Sun et al., 2013). This suggests thatmGluR5-mediated Ca2+ signals are unlikely to participate in CaN activation in intact adult brain, thereby raising the question about the mechanisms by which the activity-induced or glutamate-mediated Ca2+ elevations in adult astrocytes are generated.Another concern regards the duration and the intensity of Ca2+ transients which are very strong in experimental conditions (both in vitro and in vivo, being of mechanical, electrical or chemical nature), likely involving CICR and SOCE. It is argued that the duration ofspontaneous or evoked Ca2+ transients in healthy astrocytes both in slices (Pivneva et al., 2008; Szokolet al., 2015; Tanget al., 2015) and in intact brain (Takano et al., 2007; Kuchibhotla et al., 2009)in most cases does not exceed one minute and this would not be enough to activate CaN. However, it should be also acknowledged that CaN is sensitive to Ca2+ elevations which are as low as just a doubling of the resting [Ca2+]i , i.e., from about 70 nM to about 150 nM (Dolmetschet al., 1997; Lim et al., 2013). Yet, such Ca2+ transients might occur just in the population of astrocytes serving activated neurons. It could be that such low Ca2+ elevations are out of the performance range of the available Ca2+probes, or arerecorded as a part of noise.The tridimensional Ca2+ signalling architecture in single astrocytes in intact brain tissue is extremely complex (Volterraet al., 2014); therefore, localizedyet low amplitude Ca2+ transients may be overlooked. Finally, the CaN activation pattern within a cell appears to be not uniform (Mehtaet al., 2014),by exhibiting a steady increase in some locations and an oscillatory pattern in others, making it challenging to detect in intact brain astrocytes.
Table 2. Negative regulation of NFAT/NF-kB-mediated signaling by CaN in astrocytes.
|
Reference |
Stimulus |
Exp. time |
CaN or inhibition of activation |
Target genesor effects |
Up or Down |
|
Conboy et al., 1999 |
LPS, INFγ |
6-8 h |
Potentiation by FK506 CsA, Bafilomycin, Thapsigargin |
TNFα |
UP |
|
Pons et al., 2000 |
TNFα |
5-30 min |
Inhibition by IGF-I via CaNdephosphorylation of IkBα |
COX2 |
UP |
|
Fernandez et al., 2007 |
LPS, TNFα |
24-96 h |
Overexpresion of ΔCaNA, dephosphorylation of IkBα |
ROS, SOD |
|
|
Fernandez et al., 2007 |
Brain trauma or brain LPS |
5 days |
Overexpresion of ΔCaNA in mouse astrocytes in vivo |
Inflammatory genes including Il-1β and TNFα |
UP |
Conclusions
Over the last two decades CaN came on the stage of the astroglial (patho)physiology as a molecular switch for transforming normal astroglial phenotype into reactive astrogliosis. Being a target of a variety of stimuli, from neurotransmitters to inflammatory mediators, CaN drives transcriptional remodelling in astrocytes mainly through activation/modulation of two transcription factors, NFAT and NF-kB. While CaN directly activates NFAT, the modulation of NF-kB pathway is intricate and may result in both activation and repression of transcription. Among the transcriptional targets of astroglial CaN, there are key components of the astrocytic Ca2+signalling toolkit, astroglial marker proteins, glutamate transporters, growth factors and pro-inflammatory mediators. CaN signalling has mainly been studied in disease, while very little is known on the role of CaN in brain physiology. Detailed investigation of CaN-inducing Ca2+ signals and CaN function in astrocyte physiology is challenging but it may give a clue to our understanding of how the brain works in health and how it becomes ill, favouring the identification of pharmacological targets and novel therapeutic approaches for neurodegenerative diseases.
| Attachment | Size |
|---|---|
| 1.07 MB |
This work was supported by the Fondazione Cariplo grant 2014-1094 to DL.
The authors declare no conflict of interests.
ABDUL H.M., SAMA M.A., FURMAN J.L., MATHIS D.M., BECKETT T.L., WEIDNER A.M., PATEL E.S., BAIG I., MURPHY M.P., LEVINE H., KRANER S.D.& NORRIS C.M. (2009). Cognitive decline in Alzheimer's disease is associated with selective changes in calcineurin/NFAT signalling. The Journal of Neuroscience: The Official Journal of the Society for Neuroscience 29, 12957-12969.
ABRAHAM W.C.& BEAR M.F. (1996). Metaplasticity: the plasticity of synaptic plasticity. Trends in Neurosciences 19, 126-130.
AGULHON C., PETRAVICZ J., MCMULLEN A.B., SWEGER E.J., MINTON S.K., TAVES S.R., CASPER K.B., FIACCO T.A.& MCCARTHY K.D. (2008). What is the role of astrocyte calcium in neurophysiology? Neuron 59, 932-946.
AGULHON C., SUN M.Y., MURPHY T., MYERS T., LAUDERDALE K.& FIACCO T.A. (2012). Calcium Signaling and Gliotransmission in Normal vs. Reactive Astrocytes. Frontiers in pharmacology 3, 139.
ANNUNZIATO L., PIGNATARO G.& DI RENZO G.F. (2004). Pharmacology of brain Na+/Ca2+ exchanger: from molecular biology to therapeutic perspectives. Pharmacological reviews 56, 633-654.
ARAMBURU J., GARCIA-CÓZAR F., RAGHAVAN A., OKAMURA H., RAO A.& HOGAN P.G. (1998). Selective inhibition of NFAT activation by a peptide spanning the calcineurin targeting site of NFAT. Molecular Cell 1, 627-637.
ARAMBURU J., YAFFE M.B., LÓPEZ-RODRÍGUEZ C., CANTLEY L.C., HOGAN P.G.& RAO A. (1999). Affinity-driven peptide selection of an NFAT inhibitor more selective than cyclosporin A. Science (New York, N.Y.) 285, 2129-2133.
ARAQUE A., PARPURA V., SANZGIRI R.P.& HAYDON P.G. (1999). Tripartite synapses: glia, the unacknowledged partner. Trends in Neurosciences 22, 208-215.
ARAQUE A., CARMIGNOTO G., HAYDON P.G., OLIET S.H.R., ROBITAILLE R.& VOLTERRA A. (2014). Gliotransmitters travel in time and space. Neuron 81, 728-739.
ARONICA E., GORTER J.A., ROZEMULLER A.J., YANKAYA B.& TROOST D. (2005). Interleukin-1 beta down-regulates the expression of metabotropic glutamate receptor 5 in cultured human astrocytes. Journal of Neuroimmunology 160, 188-194.
AVNI O., LEE D., MACIAN F., SZABO S.J., GLIMCHER L.H.& RAO A. (2002). T(H) cell differentiation is accompanied by dynamic changes in histone acetylation of cytokine genes. Nature Immunology 3, 643-651.
BARAJAS M., ANDRADE A., HERNANDEZ-HERNANDEZ O., FELIX R.& ARIAS-MONTAÑO J.-A. (2008). Histamine-induced Ca2+ entry in human astrocytoma U373 MG cells: evidence for involvement of store-operated channels. Journal of Neuroscience Research 86, 3456-3468.
BAUMGÄRTEL K., GENOUX D., WELZL H., TWEEDIE-CULLEN R.Y., KOSHIBU K., LIVINGSTONE-ZATCHEJ M., MAMIE C.& MANSUY I.M. (2008). Control of the establishment of aversive memory by calcineurin and Zif268. Nature Neuroscience 11, 572-578.
BAUMGÄRTEL K.& MANSUY I.M. (2012). Neural functions of calcineurin in synaptic plasticity and memory. Learning & Memory (Cold Spring Harbor, N.Y.) 19, 375-384.
BEN HAIM L., CARRILLO-DE SAUVAGE M.-A., CEYZÉRIAT K.& ESCARTIN C. (2015). Elusive roles for reactive astrocytes in neurodegenerative diseases. Frontiers in Cellular Neuroscience 9, 278.
BENNEYWORTH M.A., LI Y., BASU A.C., BOLSHAKOV V.Y.& COYLE J.T. (2012). Cell selective conditional null mutations of serine racemase demonstrate a predominate localization in cortical glutamatergic neurons. Cellular and molecular neurobiology 32, 613-624.
BERGER J.V., DUMONT A.O., FOCANT M.C., VERGOUTS M., STERNOTTE A., CALAS A.-G., GOURSAUD S.& HERMANS E. (2012). Opposite regulation of metabotropic glutamate receptor 3 and metabotropic glutamate receptor 5 by inflammatory stimuli in cultured microglia and astrocytes. Neuroscience 205, 29-38.
BERRIDGE M.J., BOOTMAN M.D.& RODERICK H.L. (2003). Calcium signalling: dynamics, homeostasis and remodelling. Nature Reviews. Molecular Cell Biology 4, 517-529.
BHATTACHARYYA S., BIOU V., XU W., SCHLÜTER O.& MALENKA R.C. (2009). A critical role for PSD-95/AKAP interactions in endocytosis of synaptic AMPA receptors. Nature Neuroscience 12, 172-181.
BLAESER F., HO N., PRYWES R.& CHATILA T.A. (2000). Ca(2+)-dependent gene expression mediated by MEF2 transcription factors. The Journal of Biological Chemistry 275, 197-209.
BLANCO A., ALVAREZ S., FRESNO M.& MUÑOZ-FERNÁNDEZ M.A. (2008). Extracellular HIV-Tat induces cyclooxygenase-2 in glial cells through activation of nuclear factor of activated T cells. Journal of Immunology (Baltimore, Md.: 1950) 180, 530-540.
BRINI M., OTTOLINI D., CALÌ T.& CARAFOLI E. (2013). Calcium in health and disease. Metal Ions in Life Sciences 13, 81-137.
BURKARD N., BECHER J., HEINDL C., NEYSES L., SCHUH K.& RITTER O. (2005). Targeted proteolysis sustains calcineurin activation. Circulation 111, 1045-1053.
BUSHONG E.A., MARTONE M.E., JONES Y.Z.& ELLISMAN M.H. (2002). Protoplasmic astrocytes in CA1 stratum radiatum occupy separate anatomical domains. The Journal of Neuroscience: The Official Journal of the Society for Neuroscience 22, 183-192.
CAHOY J.D., EMERY B., KAUSHAL A., FOO L.C., ZAMANIAN J.L., CHRISTOPHERSON K.S., XING Y., LUBISCHER J.L., KRIEG P.A., KRUPENKO S.A., THOMPSON W.J.& BARRES B.A. (2008). A transcriptome database for astrocytes, neurons, and oligodendrocytes: a new resource for understanding brain development and function. The Journal of neuroscience : the official journal of the Society for Neuroscience 28, 264-278.
CANELLADA A., RAMIREZ B.G., MINAMI T., REDONDO J.M.& CANO E. (2008). Calcium/calcineurin signalling in primary cortical astrocyte cultures: Rcan1-4 and cyclooxygenase-2 as NFAT target genes. Glia 56, 709-722.
CARAFOLI E., GENAZZANI A.& GUERINI D. (1999). Calcium controls the transcription of its own transporters and channels in developing neurons. Biochemical and Biophysical Research Communications 266, 624-632.
CARAFOLI E., SANTELLA L., BRANCA D.& BRINI M. (2001). Generation, control, and processing of cellular calcium signals. Critical Reviews in Biochemistry and Molecular Biology 36, 107-260.
CARAFOLI E. (2004). The ambivalent nature of the calcium signal. Journal of Endocrinological Investigation 27, 134-136.
Chang C.P., McDill B.W., Neilson J.R., Joist H.E., Epstein J.A., Crabtree G.R. & Chen F. (2004) Calcineurin is required in urinary tract mesenchyme for the development of the pyeloureteral peristaltic machinery. J Clin Invest 113, 1051-1058.
CHARLES A. (2005). Reaching out beyond the synapse: glial intercellular waves coordinate metabolism. Science's STKE: signal transduction knowledge environment 270, pe6.
CHENG K.T., ONG H.L., LIU X.& AMBUDKAR I.S. (2013). Contribution and regulation of TRPC channels in store-operated Ca2+ entry. Current Topics in Membranes 71, 149-179.
CHEUNG G., SIBILLE J., ZAPATA J.& ROUACH N. (2015). Activity-Dependent Plasticity of Astroglial Potassium and Glutamate Clearance. Neural Plasticity 2015, 109106.
CLIPSTONE N.A.& CRABTREE G.R. (1992). Identification of calcineurin as a key signalling enzyme in T-lymphocyte activation. Nature 357, 695-697.
COGHLAN V.M., PERRINO B.A., HOWARD M., LANGEBERG L.K., HICKS J.B., GALLATIN W.M.& SCOTT J.D. (1995). Association of protein kinase A and protein phosphatase 2B with a common anchoring protein. Science (New York, N.Y.) 267, 108-111.
COHEN P.T., CHEN M.X.& ARMSTRONG C.G. (1996). Novel protein phosphatases that may participate in cell signalling. Advances in Pharmacology (San Diego, Calif.) 36, 67-89.
CONTRERAS L., DRAGO I., ZAMPESE E.& POZZAN T. (2010). Mitochondria: the calcium connection. Biochimica Et Biophysica Acta 1797, 607-618.
CORNELL-BELL A.H., FINKBEINER S.M., COOPER M.S.& SMITH S.J. (1990). Glutamate induces calcium waves in cultured astrocytes: long-range glial signalling. Science (New York, N.Y.) 247, 470-473.
DANI J.W., CHERNJAVSKY A.& SMITH S.J. (1992). Neuronal activity triggers calcium waves in hippocampal astrocyte networks. Neuron 8, 429-440.
DE PITTÀ M., BRUNEL N.& VOLTERRA A. (2016). Astrocytes: Orchestrating synaptic plasticity? Neuroscience 323, 43-61.
DIPOLO R.& BEAUGE L. (2006). Sodium/calcium exchanger: influence of metabolic regulation on ion carrier interactions. Physiological reviews 86, 155-203.
DISABATO D., QUAN N.& GODBOUT J.P. (2016). Neuroinflammation: The Devil is in the Details. Journal of Neurochemistry, doi: 10.1111/jnc.13607.
DOLMETSCH R.E., LEWIS R.S., GOODNOW C.C.& HEALY J.I. (1997). Differential activation of transcription factors induced by Ca2+ response amplitude and duration. Nature 386, 855-858.
Dragoni S., Laforenza U., Bonetti E., Lodola F., Bottino C., Berra-Romani R., Carlo Bongio G., Cinelli M.P., Guerra G., Pedrazzoli P., Rosti V., Tanzi F., Moccia F. (2011) Vascular endothelial growth factor stimulates endothelial colony forming cells proliferation and tubulogenesis by inducing oscillations in intracellular Ca2+ concentration. Stem Cells 29, 1898-1907.
DUPONT G., COMBETTES L., BIRD G.S.& PUTNEY J.W. (2011). Calcium oscillations. Cold Spring Harbor Perspectives in Biology 3, pii: a004226.
EL-HAGE N., BRUCE-KELLER A.J., YAKOVLEVA T., BAZOV I., BAKALKIN G., KNAPP P.E.& HAUSER K.F. (2008). Morphine exacerbates HIV-1 Tat-induced cytokine production in astrocytes through convergent effects on [Ca(2+)](i), NF-kappaB trafficking and transcription. PloS One 3, e4093.
FEDRIZZI L., LIM D.& CARAFOLI E. (2008). Calcium and signal transduction. Biochemistry and Molecular Biology Education: A Bimonthly Publication of the International Union of Biochemistry and Molecular Biology 36, 175-180.
FERNANDEZ A.M., FERNANDEZ S., CARRERO P., GARCIA-GARCIA M.& TORRES-ALEMAN I. (2007). Calcineurin in reactive astrocytes plays a key role in the interplay between proinflammatory and anti-inflammatory signals. The Journal of Neuroscience: The Official Journal of the Society for Neuroscience 27, 8745-8756.
FERNANDEZ A.M., JIMENEZ S., MECHA M., DÁVILA D., GUAZA C., VITORICA J.& TORRES-ALEMAN I. (2012). Regulation of the phosphatase calcineurin by insulin-like growth factor I unveils a key role of astrocytes in Alzheimer's pathology. Molecular Psychiatry 17, 705-718.
Fernandez A.M., Hervas R., Dominguez-Fraile M., Garrido VN., Gomez-Gutierrez P., Vega M., Vitorica J., Perez J.J., Torres Aleman I. (2016) Blockade of the Interaction of Calcineurin with FOXO in Astrocytes Protects Against Amyloid-β-Induced Neuronal Death. Journal of Alzheimers Disease 52, 1471-1478.
FIACCO T.A., AGULHON C.& MCCARTHY K.D. (2009). Sorting out astrocyte physiology from pharmacology. Annual review of pharmacology and toxicology 49, 151-174.
FILOSA J.A., NELSON M.T.& GONZALEZ BOSC L.V. (2007). Activity-dependent NFATc3 nuclear accumulation in pericytes from cortical parenchymal microvessels. American Journal of Physiology. Cell Physiology 293, C1797-1805.
FRIC J., ZELANTE T., WONG A.Y.W., MERTES A., YU H.-B.& RICCIARDI-CASTAGNOLI P. (2012). NFAT control of innate immunity. Blood 120, 1380-1389.
FRISCHBUTTER S., GABRIEL C., BENDFELDT H., RADBRUCH A.& BAUMGRASS R. (2011). Dephosphorylation of Bcl-10 by calcineurin is essential for canonical NF-κB activation in Th cells. European Journal of Immunology 41, 2349-2357.
FUJITA T., CHEN M.J., LI B., SMITH N.A., PENG W., SUN W., TONER M.J., KRESS B.T., WANG L., BENRAISS A., TAKANO T., WANG S.& NEDERGAARD M. (2014). Neuronal transgene expression in dominant-negative SNARE mice. The Journal of neuroscience : the official journal of the Society for Neuroscience 34, 16594-16604.
FURMAN J.L.& NORRIS C.M. (2014). Calcineurin and glial signalling: neuroinflammation and beyond. Journal of Neuroinflammation 11, 158.
FURMAN J.L., SAMA D.M., GANT J.C., BECKETT T.L., MURPHY M.P., BACHSTETTER A.D., VAN ELDIK L.J.& NORRIS C.M. (2012). Targeting astrocytes ameliorates neurologic changes in a mouse model of Alzheimer's disease. The Journal of neuroscience : the official journal of the Society for Neuroscience 32, 16129-16140.
FURMAN J.L., SOMPOL P., KRANER S.D., PLEISS M.M., PUTMAN E.J., DUNKERSON J., MOHMMAD ABDUL H., ROBERTS K.N., SCHEFF S.W.& NORRIS C.M. (2016). Blockade of Astrocytic Calcineurin/NFAT Signalling Helps to Normalize Hippocampal Synaptic Function and Plasticity in a Rat Model of Traumatic Brain Injury. The Journal of neuroscience : the official journal of the Society for Neuroscience 36, 1502-1515.
GARCIA-COZAR F.J., OKAMURA H., ARAMBURU J.F., SHAW K.T.Y., PELLETIER L., SHOWALTER R., VILLAFRANCA E.& RAO A. (1998). Two-site Interaction of Nuclear Factor of Activated T Cells with Activated Calcineurin. Journal of Biological Chemistry 273, 23877-23883.
GENAZZANI A.A., CARAFOLI E.& GUERINI D. (1999). Calcineurin controls inositol 1,4,5-trisphosphate type 1 receptor expression in neurons. Proceedings of the National Academy of Sciences of the United States of America 96, 5797-5801.
GLASS C.K.& OGAWA S. (2006). Combinatorial roles of nuclear receptors in inflammation and immunity. Nature Reviews. Immunology 6, 44-55.
GOLOVINA V.A. (2005). Visualization of localized store-operated calcium entry in mouse astrocytes. Close proximity to the endoplasmic reticulum. The Journal of Physiology 564, 737-749.
GOTO S., MATSUKADO Y., MIHARA Y., INOUE N.& MIYAMOTO E. (1986a). Calcineurin in human brain and its relation to extrapyramidal system. Immunohistochemical study on postmortem human brains. Acta Neuropathologica 72, 150-156.
GOTO S., MATSUKADO Y., MIHARA Y., INOUE N.& MIYAMOTO E. (1986b). Calcineurin as a neuronal marker of human brain tumors. Brain Research 371, 237-243.
GOTO S., YAMAMOTO H., FUKUNAGA K., IWASA T., MATSUKADO Y.& MIYAMOTO E. (1985). Dephosphorylation of microtubule-associated protein 2, tau factor, and tubulin by calcineurin. Journal of Neurochemistry 45, 276-283.
GRIFFITH J.P., KIM J.L., KIM E.E., SINTCHAK M.D., THOMSON J.A., FITZGIBBON M.J., FLEMING M.A., CARON P.R., HSIAO K.& NAVIA M.A. (1995). X-ray structure of calcineurin inhibited by the immunophilin-immunosuppressant FKBP12-FK506 complex. Cell 82, 507-522.
GRIMALDI M., MARATOS M.& VERMA A. (2003). Transient receptor potential channel activation causes a novel form of [Ca 2+]I oscillations and is not involved in capacitative Ca 2+ entry in glial cells. The Journal of Neuroscience: The Official Journal of the Society for Neuroscience 23, 4737-4745.
GROLLA A.A., SIM J.A., LIM D., RODRIGUEZ J.J., GENAZZANI A.A.& VERKHRATSKY A. (2013a). Amyloid-β and Alzheimer's disease type pathology differentially affects the calcium signalling toolkit in astrocytes from different brain regions. Cell Death & Disease 4, e623.
GROLLA A.A., FAKHFOURI G., BALZARETTI G., MARCELLO E., GARDONI F., CANONICO P.L., DILUCA M., GENAZZANI A.A.& LIM D. (2013b). Aβ leads to Ca²⁺ signalling alterations and transcriptional changes in glial cells. Neurobiology of Aging 34, 511-522.
GWACK Y., FESKE S., SRIKANTH S., HOGAN P.G.& RAO A. (2007). Signalling to transcription: store-operated Ca2+ entry and NFAT activation in lymphocytes. Cell Calcium 42, 145-156.
HASHIMOTO T., KAWAMATA T., SAITO N., SASAKI M., NAKAI M., NIU S., TANIGUCHI T., TERASHIMA A., YASUDA M., MAEDA K.& TANAKA C. (1998). Isoform-specific redistribution of calcineurin A alpha and A beta in the hippocampal CA1 region of gerbils after transient ischemia. Journal of Neurochemistry 70, 1289-1298.
Heit J.J., Apelqvist A.A., Gu X., Winslow M.M., Neilson J.R., Crabtree G.R. & Kim S.K. (2006) Calcineurin/NFAT signalling regulates pancreatic beta-cell growth and function. Nature 443, 345-349.
HENEKA M.T., CARSON M.J., EL KHOURY J., LANDRETH G.E., BROSSERON F., FEINSTEIN D.L., JACOBS A.H., WYSS-CORAY T., VITORICA J., RANSOHOFF R.M., HERRUP K., FRAUTSCHY S.A., FINSEN B., BROWN G.C., VERKHRATSKY A., YAMANAKA K., KOISTINAHO J., LATZ E., HALLE A., PETZOLD G.C., TOWN T., MORGAN D., SHINOHARA M.L., PERRY V.H., HOLMES C., BAZAN N.G., BROOKS D.J., HUNOT S., JOSEPH B., DEIGENDESCH N., GARASCHUK O., BODDEKE E., DINARELLO C.A., BREITNER J.C., COLE G.M., GOLENBOCK D.T.& KUMMER M.P. (2015). Neuroinflammation in Alzheimer's disease. The Lancet. Neurology 14, 388-405.
HEPPNER F.L., RANSOHOFF R.M.& BECHER B. (2015). Immune attack: the role of inflammation in Alzheimer disease. Nature Reviews. Neuroscience 16, 358-372.
HO A.M., JAIN J., RAO A.& HOGAN P.G. (1994). Expression of the transcription factor NFATp in a neuronal cell line and in the murine nervous system. The Journal of Biological Chemistry 269, 28181-28186.
HOESEL B.& SCHMID J.A. (2013). The complexity of NF-κB signalling in inflammation and cancer. Molecular Cancer 12, 86.
HOGAN P.G., CHEN L., NARDONE J.& RAO A. (2003). Transcriptional regulation by calcium, calcineurin, and NFAT. Genes & Development 17, 2205-2232.
HOLTZCLAW L.A., PANDHIT S., BARE D.J., MIGNERY G.A.& RUSSELL J.T. (2002). Astrocytes in adult rat brain express type 2 inositol 1,4,5-trisphosphate receptors. Glia 39, 69-84.
HOSOI R., MATSUDA T., ASANO S., NAKAMURA H., HASHIMOTO H., TAKUMA K.& BABA A. (1997). Isoform-specific up-regulation by ouabain of Na+,K+-ATPase in cultured rat astrocytes. Journal of Neurochemistry 69, 2189-2196.
HOTH M., BUTTON D.C.& LEWIS R.S. (2000). Mitochondrial control of calcium-channel gating: a mechanism for sustained signalling and transcriptional activation in T lymphocytes. Proceedings of the National Academy of Sciences of the United States of America 97, 10607-10612.
HUAI Q., KIM H.-Y., LIU Y., ZHAO Y., MONDRAGON A., LIU J.O.& KE H. (2002). Crystal structure of calcineurin-cyclophilin-cyclosporin shows common but distinct recognition of immunophilin-drug complexes. Proceedings of the National Academy of Sciences of the United States of America 99, 12037-12042.
HUANG W., FILETA J., RAWE I., QU J.& GROSSKREUTZ C.L. (2010). Calpain activation in experimental glaucoma. Investigative Ophthalmology & Visual Science 51, 3049-3054.
HUDSON M.B.& PRICE S.R. (2013). Calcineurin: a poorly understood regulator of muscle mass. The International Journal of Biochemistry & Cell Biology 45, 2173-2178.
ILLES P., VERKHRATSKY A., BURNSTOCK G.& FRANKE H. (2012). P2X receptors and their roles in astroglia in the central and peripheral nervous system. The Neuroscientist : a review journal bringing neurobiology, neurology and psychiatry 18, 422-438.
IM S.-H.& RAO A. (2004). Activation and deactivation of gene expression by Ca2+/calcineurin-NFAT-mediated signalling. Molecules and Cells 18, 1-9.
INGEBRITSEN T.S.& COHEN P. (1983). The Protein Phosphatases Involved in Cellular Regulation. 1. Classification and Substrate Specificities. European Journal of Biochemistry 132, 255-261.
JAIN J., MCCAFFREY P.G., MINER Z., KERPPOLA T.K., LAMBERT J.N., VERDINE G.L., CURRAN T.& RAO A. (1993). The T-cell transcription factor NFATp is a substrate for calcineurin and interacts with Fos and Jun. Nature 365, 352-355.
JAMES G.& BUTT A.M. (2002). P2Y and P2X purinoceptor mediated Ca2+ signalling in glial cell pathology in the central nervous system. European Journal of Pharmacology 447, 247-260.
JIN S.M., CHO H.J., KIM Y.W., HWANG J.Y.& MOOK-JUNG I. (2012). Aβ-induced Ca(2+) influx regulates astrocytic BACE1 expression via calcineurin/NFAT4 signals. Biochemical and Biophysical Research Communications 425, 649-655.
JONES E.A., SUN D., KOBIERSKI L.& SYMES A.J. (2003). NFAT4 is expressed in primary astrocytes and activated by glutamate. Journal of Neuroscience Research 72, 191-197.
KAKALIS L.T., KENNEDY M., SIKKINK R., RUSNAK F.& ARMITAGE I.M. (1995). Characterization of the calcium-binding sites of calcineurin B. FEBS letters 362, 55-58.
KAMINSKA B., GAWEDA-WALERYCH K.& ZAWADZKA M. (2004). Molecular mechanisms of neuroprotective action of immunosuppressants--facts and hypotheses. Journal of Cellular and Molecular Medicine 8, 45-58.
KIMELBERG H.K. (2007). Supportive or information-processing functions of the mature protoplasmic astrocyte in the mammalian CNS? A critical appraisal. Neuron glia biology 3, 181-189.
KIRISCHUK S., PARPURA V.& VERKHRATSKY A. (2012). Sodium dynamics: another key to astroglial excitability? Trends in Neurosciences 35, 497-506.
KISSINGER C.R., PARGE H.E., KNIGHTON D.R., LEWIS C.T., PELLETIER L.A., TEMPCZYK A., KALISH V.J., TUCKER K.D., SHOWALTER R.E.& MOOMAW E.W. (1995). Crystal structures of human calcineurin and the human FKBP12-FK506-calcineurin complex. Nature 378, 641-644.
KLEE C.B., CROUCH T.H.& KRINKS M.H. (1979). Calcineurin: a calcium- and calmodulin-binding protein of the nervous system. Proceedings of the National Academy of Sciences of the United States of America 76, 6270-6273.
KLEE C.B., DRAETTA G.F.& HUBBARD M.J. (1988). Calcineurin. Advances in Enzymology and Related Areas of Molecular Biology 61, 149-200.
KLEE C.B., REN H.& WANG X. (1998). Regulation of the Calmodulin-stimulated Protein Phosphatase, Calcineurin. Journal of Biological Chemistry 273, 13367-13370.
KOIZUMI S. (2010). Synchronization of Ca2+ oscillations: involvement of ATP release in astrocytes. The FEBS journal 277, 286-292.
KÖNIG N., RAYNAUD F., FEANE H., DURAND M., MESTRE-FRANCÈS N., ROSSEL M., OUALI A.& BENYAMIN Y. (2003). Calpain 3 is expressed in astrocytes of rat and Microcebus brain. Journal of Chemical Neuroanatomy 25, 129-136.
KRAMER D., FRESU L., ASHBY D.S., FREEMAN T.C.& GENAZZANI A.A. (2003). Calcineurin controls the expression of numerous genes in cerebellar granule cells. Molecular and Cellular Neurosciences 23, 325-330.
KUCHIBHOTLA K.V., LATTARULO C.R., HYMAN B.T.& BACSKAI B.J. (2009). Synchronous hyperactivity and intercellular calcium waves in astrocytes in Alzheimer mice. Science 323, 1211-1215.
KUNO T., MUKAI H., ITO A., CHANG C.D., KISHIMA K., SAITO N.& TANAKA C. (1992). Distinct cellular expression of calcineurin A alpha and A beta in rat brain. Journal of Neurochemistry 58, 1643-1651.
LAKSHMIKUTTYAMMA A., SELVAKUMAR P., SHARMA A.R., ANDERSON D.H.& SHARMA R.K. (2004). In vitro proteolytic degradation of bovine brain calcineurin by m-calpain. Neurochemical Research 29, 1913-1921.
LAKSHMIKUTTYAMMA A., SELVAKUMAR P., KAKKAR R., KANTHAN R., WANG R.& SHARMA R.K. (2003). Activation of calcineurin expression in ischemia-reperfused rat heart and in human ischemic myocardium. Journal of Cellular Biochemistry 90, 987-997.
LEE S.D., COLLA E., SHEEN M.R., NA K.Y.& KWON H.M. (2003). Multiple domains of TonEBP cooperate to stimulate transcription in response to hypertonicity. The Journal of Biological Chemistry 278, 47571-47577.
LEE Y.B., DU S., RHIM H., LEE E.B., MARKELONIS G.J.& OH T.H. (2000). Rapid increase in immunoreactivity to GFAP in astrocytes in vitro induced by acidic pH is mediated by calcium influx and calpain I. Brain Research 864, 220-229.
LIEBERMAN D.N.& MODY I. (1994). Regulation of NMDA channel function by endogenous Ca(2+)-dependent phosphatase. Nature 369, 235-239.
LIM D., RONCO V., GROLLA A.A., VERKHRATSKY A.& GENAZZANI A.A. (2014). Glial calcium signalling in Alzheimer's disease. Reviews of Physiology, Biochemistry and Pharmacology 167, 45-65.
LIM D., IYER A., RONCO V., GROLLA A.A., CANONICO P.L., ARONICA E.& GENAZZANI A.A. (2013). Amyloid beta deregulates astroglial mGluR5-mediated calcium signalling via calcineurin and Nf-kB. Glia 61, 1134-1145.
LIM D., RODRÍGUEZ-ARELLANO J.J., PARPURA V., ZOREC R., ZEIDÁN-CHULIÁ F., GENAZZANI A.A.& VERKHRATSKY A. (2016). Calcium signalling toolkits in astrocytes and spatio-temporal progression of Alzheimer's disease. Current Alzheimer Research 13, 359-369.
LIN S.C.& BERGLES D.E. (2002). Physiological characteristics of NG2-expressing glial cells. Journal of neurocytology 31, 537-549.
LIND B.L., BRAZHE A.R., JESSEN S.B., TAN F.C.C.& LAURITZEN M.J. (2013). Rapid stimulus-evoked astrocyte Ca2+ elevations and hemodynamic responses in mouse somatosensory cortex in vivo. Proceedings of the National Academy of Sciences of the United States of America 110, E4678-4687.
LIU F., GRUNDKE-IQBAL I., IQBAL K., ODA Y., TOMIZAWA K.& GONG C.-X. (2005). Truncation and activation of calcineurin A by calpain I in Alzheimer disease brain. The Journal of Biological Chemistry 280, 37755-37762.
LIU J.S., JOHN G.R., SIKORA A., LEE S.C.& BROSNAN C.F. (2000). Modulation of interleukin-1beta and tumor necrosis factor alpha signalling by P2 purinergic receptors in human fetal astrocytes. The Journal of Neuroscience: The Official Journal of the Society for Neuroscience 20, 5292-5299.
LODYGIN D., ODOARDI F., SCHLAGER C., KORNER H., KITZ A., NOSOV M., VAN DEN BRANDT J., REICHARDT H.M., HABERL M.& FLUGEL A. (2013). A combination of fluorescent NFAT and H2B sensors uncovers dynamics of T cell activation in real time during CNS autoimmunity. Nature medicine 19, 784-790.
MACIÁN F., GARCÍA-CÓZAR F., IM S.-H., HORTON H.F., BYRNE M.C.& RAO A. (2002). Transcriptional mechanisms underlying lymphocyte tolerance. Cell 109, 719-731.
Maillet M., Davis J., Auger-Messier M., York A., Osinska H., Piquereau J., Lorenz J.N., Robbins J., Ventura-Clapier R. & Molkentin J.D. (2010) Heart-specific deletion of CnB1 reveals multiple mechanisms whereby calcineurin regulates cardiac growth and function. J Biol Chem 285, 6716-6724.
MALARKEY E.B., NI Y.& PARPURA V. (2008). Ca2+ entry through TRPC1 channels contributes to intracellular Ca2+ dynamics and consequent glutamate release from rat astrocytes. Glia 56, 821-835.
MALLINSON J., MEISSNER J.& CHANG K.-C. (2009). Chapter 2. Calcineurin signalling and the slow oxidative skeletal muscle fiber type. International Review of Cell and Molecular Biology 277, 67-101.
MARTINEZ-MARTINEZ S.& REDONDO J.M. (2004). Inhibitors of the calcineurin/NFAT pathway. Current medicinal chemistry 11, 997-1007.
MARTÍNEZ-MARTÍNEZ S., RODRÍGUEZ A., LÓPEZ-MADERUELO M.D., ORTEGA-PÉREZ I., VÁZQUEZ J.& REDONDO J.M. (2006). Blockade of NFAT activation by the second calcineurin binding site. The Journal of Biological Chemistry 281, 6227-6235.
MATSUDA T., TAKUMA K., ASANO S., KISHIDA Y., NAKAMURA H., MORI K., MAEDA S.& BABA A. (1998). Involvement of calcineurin in Ca2+ paradox-like injury of cultured rat astrocytes. Journal of Neurochemistry 70, 2004-2011.
MCKINSEY T.A., ZHANG C.L.& OLSON E.N. (2000). Activation of the myocyte enhancer factor-2 transcription factor by calcium/calmodulin-dependent protein kinase-stimulated binding of 14-3-3 to histone deacetylase 5. Proceedings of the National Academy of Sciences of the United States of America 97, 14400-14405.
MEHTA S., AYE-HAN N.N., GANESAN A., OLDACH L., GORSHKOV K.& ZHANG J. (2014). Calmodulin-controlled spatial decoding of oscillatory Ca2+ signals by calcineurin. eLife 3, e03765.
MOCCIA F., ZUCCOLO E., SODA T., TANZI F., GUERRA G., MAPELLI L., LODOLA F.& D'ANGELO E. (2015). Stim and Orai proteins in neuronal Ca(2+) signalling and excitability. Frontiers in Cellular Neuroscience 9, 153.
MOHMMAD ABDUL H., BAIG I., LEVINE H., GUTTMANN R.P.& NORRIS C.M. (2011). Proteolysis of calcineurin is increased in human hippocampus during mild cognitive impairment and is stimulated by oligomeric Abeta in primary cell culture. Aging Cell 10, 103-113.
MULKEY R.M., ENDO S., SHENOLIKAR S.& MALENKA R.C. (1994). Involvement of a calcineurin/inhibitor-1 phosphatase cascade in hippocampal long-term depression. Nature 369, 486-488.
MURAMATSU T.& KINCAID R.L. (1992). Molecular cloning and chromosomal mapping of the human gene for the testis-specific catalytic subunit of calmodulin-dependent protein phosphatase (calcineurin A). Biochemical and Biophysical Research Communications 188, 265-271.
NATH A., CONANT K., CHEN P., SCOTT C.& MAJOR E.O. (1999). Transient exposure to HIV-1 Tat protein results in cytokine production in macrophages and astrocytes. A hit and run phenomenon. The Journal of Biological Chemistry 274, 17098-17102.
NEDERGAARD M., RANSOM B.& GOLDMAN S.A. (2003). New roles for astrocytes: redefining the functional architecture of the brain. Trends in Neurosciences 26, 523-530.
NEDERGAARD M.& VERKHRATSKY A. (2012). Artifact versus reality--how astrocytes contribute to synaptic events. Glia 60, 1013-1023.
NEGULESCU P.A., SHASTRI N.& CAHALAN M.D. (1994). Intracellular calcium dependence of gene expression in single T lymphocytes. Proceedings of the National Academy of Sciences of the United States of America 91, 2873-2877.
NERIA F., DEL CARMEN SERRANO-PEREZ M., VELASCO P., URSO K., TRANQUE P.& CANO E. (2013). NFATc3 promotes Ca(2+) -dependent MMP3 expression in astroglial cells. Glia 61, 1052-1066.
NEWMAN R.H.& ZHANG J. (2008). Visualization of phosphatase activity in living cells with a FRET-based calcineurin activity sensor. Molecular bioSystems 4, 496-501.
NEWTON K.& DIXIT V.M. (2012). Signalling in innate immunity and inflammation. Cold Spring Harbor Perspectives in Biology 4, pii: a006049.
NORRIS C.M., KADISH I., BLALOCK E.M., CHEN K.-C., THIBAULT V., PORTER N.M., LANDFIELD P.W.& KRANER S.D. (2005). Calcineurin triggers reactive/inflammatory processes in astrocytes and is upregulated in aging and Alzheimer's models. The Journal of Neuroscience: The Official Journal of the Society for Neuroscience 25, 4649-4658.
OH-HORA M.& RAO A. (2008). Calcium signalling in lymphocytes. Current Opinion in Immunology 20, 250-258.
OIKAWA H., NAKAMICHI N., KAMBE Y., OGURA M.& YONEDA Y. (2005). An increase in intracellular free calcium ions by nicotinic acetylcholine receptors in a single cultured rat cortical astrocyte. Journal of neuroscience research 79, 535-544.
OKUBO Y., BLAKESLEY V.A., STANNARD B., GUTKIND S.& LE ROITH D. (1998). Insulin-like growth factor-I inhibits the stress-activated protein kinase/c-Jun N-terminal kinase. The Journal of Biological Chemistry 273, 25961-25966.
OLIVEIRA J.F., SARDINHA V.M., GUERRA-GOMES S., ARAQUE A.& SOUSA N. (2015). Do stars govern our actions? Astrocyte involvement in rodent behavior. Trends in Neurosciences 38, 535-549.
ORRE M., KAMPHUIS W., OSBORN L.M., JANSEN A.H., KOOIJMAN L., BOSSERS K.& HOL E.M. (2014). Isolation of glia from Alzheimer's mice reveals inflammation and dysfunction. Neurobiology of aging 35, 2746-2760.
OTA Y., ZANETTI A.T.& HALLOCK R.M. (2013). The role of astrocytes in the regulation of synaptic plasticity and memory formation. Neural Plasticity 2013, 185463.
PALKOWITSCH L., MARIENFELD U., BRUNNER C., EITELHUBER A., KRAPPMANN D.& MARIENFELD R.B. (2011). The Ca2+-dependent phosphatase calcineurin controls the formation of the Carma1-Bcl10-Malt1 complex during T cell receptor-induced NF-kappaB activation. The Journal of Biological Chemistry 286, 7522-7534.
PALLEN C.J.& WANG J.H. (1985). A multifunctional calmodulin-stimulated phosphatase. Archives of Biochemistry and Biophysics 237, 281-291.
PALYGIN O., LALO U., VERKHRATSKY A.& PANKRATOV Y. (2010). Ionotropic NMDA and P2X1/5 receptors mediate synaptically induced Ca2+ signalling in cortical astrocytes. Cell Calcium 48, 225-231.
PAN M.-G., XIONG Y.& CHEN F. (2013). NFAT gene family in inflammation and cancer. Current Molecular Medicine 13, 543-554.
PAREKH A.B. (2008). Ca2+ microdomains near plasma membrane Ca2+ channels: impact on cell function. The Journal of Physiology 586, 3043-3054.
PAREKH A.B. (2011). Decoding cytosolic Ca2+ oscillations. Trends in Biochemical Sciences 36, 78-87.
PARPURA V.& HAYDON P.G. (2000). Physiological astrocytic calcium levels stimulate glutamate release to modulate adjacent neurons. Proceedings of the National Academy of Sciences of the United States of America 97, 8629-8634.
Parsons S.A., Millay D.P., Sargent M.A., Naya F.J., McNally E.M., Sweeney H.L. & Molkentin J.D. (2007) Genetic disruption of calcineurin improves skeletal muscle pathology and cardiac disease in a mouse model of limb-girdle muscular dystrophy. J Biol Chem 282, 10068-10078.
PAUL S.& SCHAEFER B.C. (2013). A new look at T cell receptor signalling to nuclear factor-κB. Trends in Immunology 34, 269-281.
PEKNY M.& PEKNA M. (2014). Astrocyte reactivity and reactive astrogliosis: costs and benefits. Physiological Reviews 94, 1077-1098.
PEREA G., SUR M.& ARAQUE A. (2014). Neuron-glia networks: integral gear of brain function. Frontiers in Cellular Neuroscience 8, 378.
PEREIRA A.& FURLAN F.A. (2010). Astrocytes and human cognition: modeling information integration and modulation of neuronal activity. Progress in Neurobiology 92, 405-420.
PÉREZ-ORTIZ J.M., SERRANO-PÉREZ M.C., PASTOR M.D., MARTÍN E.D., CALVO S., RINCÓN M.& TRANQUE P. (2008). Mechanical lesion activates newly identified NFATc1 in primary astrocytes: implication of ATP and purinergic receptors. The European Journal of Neuroscience 27, 2453-2465.
PERLMUTTER L.S., GALL C., BAUDRY M.& LYNCH G. (1990). Distribution of calcium-activated protease calpain in the rat brain. The Journal of Comparative Neurology 296, 269-276.
PETRAVICZ J., FIACCO T.A.& MCCARTHY K.D. (2008). Loss of IP3 receptor-dependent Ca2+ increases in hippocampal astrocytes does not affect baseline CA1 pyramidal neuron synaptic activity. The Journal of Neuroscience: The Official Journal of the Society for Neuroscience 28, 4967-4973.
PIRTTIMAKI T.M.& PARRI H.R. (2013). Astrocyte plasticity: implications for synaptic and neuronal activity. The Neuroscientist: A Review Journal Bringing Neurobiology, Neurology and Psychiatry 19, 604-615.
PIVNEVA T., HAAS B., REYES-HARO D., LAUBE G., VEH R.W., NOLTE C., SKIBO G.& KETTENMANN H. (2008). Store-operated Ca2+ entry in astrocytes: different spatial arrangement of endoplasmic reticulum explains functional diversity in vitro and in situ. Cell Calcium 43, 591-601.
PIZZO P., BURGO A., POZZAN T.& FASOLATO C. (2001). Role of capacitative calcium entry on glutamate-induced calcium influx in type-I rat cortical astrocytes. Journal of Neurochemistry 79, 98-109.
PONS S.& TORRES-ALEMAN I. (2000). Insulin-like growth factor-I stimulates dephosphorylation of ikappa B through the serine phosphatase calcineurin (protein phosphatase 2B). The Journal of Biological Chemistry 275, 38620-38625.
PUTNEY J.W. (2007). New molecular players in capacitative Ca2+ entry. Journal of Cell Science 120, 1959-1965.
RANDRIAMAMPITA C.& TSIEN R.Y. (1995). Degradation of a calcium influx factor (CIF) can be blocked by phosphatase inhibitors or chelation of Ca2+. The Journal of Biological Chemistry 270, 29-32.
REESE L.C.& TAGLIALATELA G. (2010). Neuroimmunomodulation by calcineurin in aging and Alzheimer's disease. Aging and Disease 1, 245-253.
REESE L.C.& TAGLIALATELA G. (2011). A role for calcineurin in Alzheimer's disease. Current Neuropharmacology 9, 685-692.
REYES R.C., VERKHRATSKY A.& PARPURA V. (2012). Plasmalemmal Na+/Ca2+ exchanger modulates Ca2+-dependent exocytotic release of glutamate from rat cortical astrocytes. ASN neuro 4, pii: e00075.
RIZZUTO R., PINTON P., BRINI M., CHIESA A., FILIPPIN L.& POZZAN T. (1999). Mitochondria as biosensors of calcium microdomains. Cell Calcium 26, 193-199.
RONCO V., GROLLA A.A., GLASNOV T.N., CANONICO P.L., VERKHRATSKY A., GENAZZANI A.A.& LIM D. (2014). Differential deregulation of astrocytic calcium signalling by amyloid-β, TNFα, IL-1β and LPS. Cell Calcium 55, 219-229.
ROSEBECK S., REHMAN A.O., LUCAS P.C.& MCALLISTER-LUCAS L.M. (2011). From MALT lymphoma to the CBM signalosome: three decades of discovery. Cell Cycle (Georgetown, Tex.) 10, 2485-2496.
SADIGH-ETEGHAD S., MAJDI A., MAHMOUDI J., GOLZARI S.E.& TALEBI M. (2016). Astrocytic and microglial nicotinic acetylcholine receptors: an overlooked issue in Alzheimer's disease. Journal of neural transmission.
SALIDO G.M., SAGE S.O.& ROSADO J.A. (2009). Biochemical and functional properties of the store-operated Ca2+ channels. Cell Signal 21, 457-461.
SAMA M.A., MATHIS D.M., FURMAN J.L., ABDUL H.M., ARTIUSHIN I.A., KRANER S.D.& NORRIS C.M. (2008). Interleukin-1beta-dependent signalling between astrocytes and neurons depends critically on astrocytic calcineurin/NFAT activity. The Journal of Biological Chemistry 283, 21953-21964.
SANTELLA L., ERCOLANO E., LIM D., NUSCO G.A.& MOCCIA F. (2003). Activated M-phase-promoting factor (MPF) is exported from the nucleus of starfish oocytes to increase the sensitivity of the Ins(1,4,5)P3 receptors. Biochemical Society Transactions 31, 79-82.
SCEMES E.& GIAUME C. (2006). Astrocyte calcium waves: what they are and what they do. Glia 54, 716-725.
SCHIAFFINO S. (2010). Fibre types in skeletal muscle: a personal account. Acta Physiologica (Oxford, England) 199, 451-463.
SCHUBERT P., MORINO T., MIYAZAKI H., OGATA T., NAKAMURA Y., MARCHINI C.& FERRONI S. (2000). Cascading glia reactions: a common pathomechanism and its differentiated control by cyclic nucleotide signaling. Annals of the New York Academy of Sciences 903, 24-33.
SCHWANINGER M., SALLMANN S., PETERSEN N., SCHNEIDER A., PRINZ S., LIBERMANN T.A.& SPRANGER M. (1999). Bradykinin induces interleukin-6 expression in astrocytes through activation of nuclear factor-kappaB. Journal of Neurochemistry 73, 1461-1466.
SELKOE D.J.& HARDY J. (2016). The amyloid hypothesis of Alzheimer's disease at 25 years. EMBO molecular medicine, pii: e201606210.
SERRANO-PÉREZ M.C., MARTÍN E.D., VAQUERO C.F., AZCOITIA I., CALVO S., CANO E.& TRANQUE P. (2011). Response of transcription factor NFATc3 to excitotoxic and traumatic brain insults: identification of a subpopulation of reactive astrocytes. Glia 59, 94-107.
SHAO Y.& MCCARTHY K.D. (1994). Plasticity of astrocytes. Glia 11, 147-155.
SHAW J.P., UTZ P.J., DURAND D.B., TOOLE J.J., EMMEL E.A.& CRABTREE G.R. (1988). Identification of a putative regulator of early T cell activation genes. Science (New York, N.Y.) 241, 202-205.
SHIGETOMI E., PATEL S.& KHAKH B.S. (2016). Probing the Complexities of Astrocyte Calcium Signalling. Trends in Cell Biology 26, 300-312.
SHIH V.F.-S., TSUI R., CALDWELL A.& HOFFMANN A. (2011). A single NFκB system for both canonical and non-canonical signalling. Cell Research 21, 86-102.
SHIODA N., MORIGUCHI S., SHIRASAKI Y.& FUKUNAGA K. (2006). Generation of constitutively active calcineurin by calpain contributes to delayed neuronal death following mouse brain ischemia. Journal of Neurochemistry 98, 310-320.
SLOAN S.A.& BARRES B.A. (2014). Looks can be deceiving: reconsidering the evidence for gliotransmission. Neuron 84, 1112-1115.
SMEDLER E.& UHLÉN P. (2014). Frequency decoding of calcium oscillations. Biochimica Et Biophysica Acta 1840, 964-969.
SOBRADO M., RAMIREZ B.G., NERIA F., LIZASOAIN I., ARBONES M.L., MINAMI T., REDONDO J.M., MORO M.A.& CANO E. (2012). Regulator of calcineurin 1 (Rcan1) has a protective role in brain ischemia/reperfusion injury. Journal of Neuroinflammation 9, 48.
STEMMER P.M.& KLEE C.B. (1994). Dual calcium ion regulation of calcineurin by calmodulin and calcineurin B. Biochemistry 33, 6859-6866.
STRAUB S.V., GIOVANNUCCI D.R.& YULE D.I. (2000). Calcium wave propagation in pancreatic acinar cells: functional interaction of inositol 1,4,5-trisphosphate receptors, ryanodine receptors, and mitochondria. The Journal of General Physiology 116, 547-560.
SUN W., MCCONNELL E., PARE J.F., XU Q., CHEN M., PENG W., LOVATT D., HAN X., SMITH Y.& NEDERGAARD M. (2013). Glutamate-dependent neuroglial calcium signaling differs between young and adult brain. Science 339, 197-200.
SZOKOL K., HEUSER K., TANG W., JENSEN V., ENGER R., BEDNER P., STEINHAUSER C., TAUBOLL E., OTTERSEN O.P.& NAGELHUS E.A. (2015). Augmentation of Ca(2+) signaling in astrocytic endfeet in the latent phase of temporal lobe epilepsy. Frontiers in cellular neuroscience 9, 49.
TAKANO T., HAN X., DEANE R., ZLOKOVIC B.& NEDERGAARD M. (2007). Two-photon imaging of astrocytic Ca2+ signaling and the microvasculature in experimental mice models of Alzheimer's disease. Annals of the New York Academy of Sciences 1097, 40-50.
TAKANO T., HE W., HAN X., WANG F., XU Q., WANG X., OBERHEIM BUSH N.A., CRUZ N., DIENEL G.A.& NEDERGAARD M. (2014). Rapid manifestation of reactive astrogliosis in acute hippocampal brain slices. Glia 62, 78-95.
TAKUMA K., MATSUDA T., HASHIMOTO H., ASANO S.& BABA A. (1994). Cultured rat astrocytes possess Na(+)-Ca2+ exchanger. Glia 12, 336-342.
TAKUMA K., LEE E., KIDAWARA M., MORI K., KIMURA Y., BABA A.& MATSUDA T. (1999). Apoptosis in Ca2 + reperfusion injury of cultured astrocytes: roles of reactive oxygen species and NF-kappaB activation. The European Journal of Neuroscience 11, 4204-4212.
TAKUMA K., BABA A.& MATSUDA T. (2004). Astrocyte apoptosis: implications for neuroprotection. Progress in Neurobiology 72, 111-127.
TALLANT E.A., BRUMLEY L.M.& WALLACE R.W. (1988). Activation of a calmodulin-dependent phosphatase by a Ca2+-dependent protease. Biochemistry 27, 2205-2211.
TANG W., SZOKOL K., JENSEN V., ENGER R., TRIVEDI C.A., HVALBY O., HELM P.J., LOOGER L.L., SPRENGEL R.& NAGELHUS E.A. (2015). Stimulation-evoked Ca2+ signals in astrocytic processes at hippocampal CA3-CA1 synapses of adult mice are modulated by glutamate and ATP. The Journal of neuroscience : the official journal of the Society for Neuroscience 35, 3016-3021.
TEAKTONG T., GRAHAM A., COURT J., PERRY R., JAROS E., JOHNSON M., HALL R.& PERRY E. (2003). Alzheimer's disease is associated with a selective increase in alpha7 nicotinic acetylcholine receptor immunoreactivity in astrocytes. Glia 41, 207-211.
THEODOSIS D.T.& POULAIN D.A. (1999). Contribution of astrocytes to activity-dependent structural plasticity in the adult brain. Adv Exp Med Biol 468, 175-182.
THEODOSIS D.T., POULAIN D.A.& OLIET S.H.R. (2008). Activity-dependent structural and functional plasticity of astrocyte-neuron interactions. Physiological Reviews 88, 983-1008.
TILLEUX S., BERGER J.& HERMANS E. (2007). Induction of astrogliosis by activated microglia is associated with a down-regulation of metabotropic glutamate receptor 5. Journal of Neuroimmunology 189, 23-30.
TUSCHICK S., KIRISCHUK S., KIRCHHOFF F., LIEFELDT L., PAUL M., VERKHRATSKY A.& KETTENMANN H. (1997). Bergmann glial cells in situ express endothelinB receptors linked to cytoplasmic calcium signals. Cell Calcium 21, 409-419.
UHLEN P.& FRITZ N. (2010). Biochemistry of calcium oscillations. Biochem Biophys Res Commun 396, 28-32.
VERKHRATSKY A.& KETTENMANN H. (1996). Calcium signalling in glial cells. Trends in Neurosciences 19, 346-352.
VERKHRATSKY A.& STEINHAUSER C. (2000). Ion channels in glial cells. Brain research. Brain research reviews 32, 380-412.
VERKHRATSKY A.& KIRCHHOFF F. (2007). NMDA Receptors in glia. The Neuroscientist : a review journal bringing neurobiology, neurology and psychiatry 13, 28-37.
VERKHRATSKY A., RODRÍGUEZ J.J.& PARPURA V. (2012a). Calcium signalling in astroglia. Molecular and Cellular Endocrinology 353, 45-56.
VERKHRATSKY A., PANKRATOV Y., LALO U.& NEDERGAARD M. (2012b). P2X receptors in neuroglia. Wiley Interdisciplinary Reviews. Membrane Transport and Signalling 1, doi: 10.1002/wmts.12.
VERKHRATSKY A.& NEDERGAARD M. (2014). Astroglial cradle in the life of the synapse. Philosophical transactions of the Royal Society of London. Series B, Biological sciences 369, 20130595.
VERKHRATSKY A.& PARPURA V. (2014). Store-operated calcium entry in neuroglia. Neuroscience Bulletin 30, 125-133.
VERKHRATSKY A., REYES R.C.& PARPURA V. (2014). TRP channels coordinate ion signalling in astroglia. Reviews of Physiology, Biochemistry and Pharmacology 166, 1-22.
VERKHRATSKY A.B., ARTHUR MORGAN (2007). Glial Neurobiology, Wiley-Blackwell, 230 pp.
VINADÉ L., GONÇALVES C.A., WOFCHUK S., GOTTFRIED C.& RODNIGHT R. (1997). Evidence for a role for calcium ions in the dephosphorylation of glial fibrillary acidic protein (GFAP) in immature hippocampal slices and in astrocyte cultures from the rat. Brain Research. Developmental Brain Research 104, 11-17.
VOLTERRA A., LIAUDET N.& SAVTCHOUK I. (2014). Astrocyte Ca²⁺ signalling: an unexpected complexity. Nature Reviews. Neuroscience 15, 327-335.
WALSH C., BARROW S., VORONINA S., CHVANOV M., PETERSEN O.H.& TEPIKIN A. (2009). Modulation of calcium signalling by mitochondria. Biochim Biophys Acta 1787, 1374-1382.
WANG H.G., PATHAN N., ETHELL I.M., KRAJEWSKI S., YAMAGUCHI Y., SHIBASAKI F., MCKEON F., BOBO T., FRANKE T.F.& REED J.C. (1999). Ca2+-induced apoptosis through calcineurin dephosphorylation of BAD. Science (New York, N.Y.) 284, 339-343.
WANG J.H.& DESAI R. (1976). A brain protein and its effect on the Ca2+-and protein modulator-activated cyclic nucleotide phosphodiesterase. Biochemical and Biophysical Research Communications 72, 926-932.
WANG K.K., ROUFOGALIS B.D.& VILLALOBO A. (1989). Characterization of the fragmented forms of calcineurin produced by calpain I. Biochemistry and Cell Biology = Biochimie Et Biologie Cellulaire 67, 703-711.
WU H.-Y., TOMIZAWA K.& MATSUI H. (2007). Calpain-calcineurin signalling in the pathogenesis of calcium-dependent disorder. Acta Medica Okayama 61, 123-137.
WU H.-Y., TOMIZAWA K., ODA Y., WEI F.-Y., LU Y.-F., MATSUSHITA M., LI S.-T., MORIWAKI A.& MATSUI H. (2004). Critical role of calpain-mediated cleavage of calcineurin in excitotoxic neurodegeneration. The Journal of Biological Chemistry 279, 4929-4940.
YAN H.Q., SHIN S.S., MA X., LI Y.& DIXON C.E. (2014). Differential effect of traumatic brain injury on the nuclear factor of activated T Cells C3 and C4 isoforms in the rat hippocampus. Brain Research 1548, 63-72.
YANEZ M., GIL-LONGO J.& CAMPOS-TOIMIL M. (2012). Calcium binding proteins. Advances in experimental medicine and biology 740, 461-482.
YANG S.A.& KLEE C.B. (2000). Low affinity Ca2+-binding sites of calcineurin B mediate conformational changes in calcineurin A. Biochemistry 39, 16147-16154.
ZENG H., CHATTARJI S., BARBAROSIE M., RONDI-REIG L., PHILPOT B.D., MIYAKAWA T., BEAR M.F.& TONEGAWA S. (2001). Forebrain-specific calcineurin knockout selectively impairs bidirectional synaptic plasticity and working/episodic-like memory. Cell 107, 617-629.
ZHANG Y., CHEN K., SLOAN S.A., BENNETT M.L., SCHOLZE A.R., O'KEEFFE S., PHATNANI H.P., GUARNIERI P., CANEDA C., RUDERISCH N., DENG S., LIDDELOW S.A., ZHANG C., DANEMAN R., MANIATIS T., BARRES B.A.& WU J.Q. (2014). An RNA-sequencing transcriptome and splicing database of glia, neurons, and vascular cells of the cerebral cortex. The Journal of neuroscience : the official journal of the Society for Neuroscience 34, 11929-11947.
ZONTA M.& CARMIGNOTO G. (2002). Calcium oscillations encoding neuron-to-astrocyte communication. Journal of Physiology, Paris 96, 193-198.