Neurogenesis is one of the most striking phenomena in the adult rodent brain, however, it is limited to only particular brain regions including the olfactory bulb and the dentate gyrus (Altman, 1962; Altman, 1963). In the subventricular zone (SVZ) newborn neurons are produced and travel a long distance through the rostral migratory stream to reach the OB where they become diverse types of interneurons (Doetsch and Alvarez-Buylla, 1996; Doetsch et al., 1999; Lim and Alvarez-Buylla, 2016). In contrast, the neural stem cells located in the subgranular zone (SGZ) of the dentate gyrus generate newborn neurons that integrate locally into the granule cell layer (Altman and Das, 1965; Altman and Bayer, 1990; Curtis et al., 2012). Neurogenesis in the dentate was also described in birds (Goldman and Nottebohm, 1983) and other species, including non-human primates (Gould et al., 1999) and even humans (Eriksson et al., 1998) although recent studies have cast doubt on this in adult humans (Sorrels et al., 2018). The exact function of DG neurogenesis is still unknown but reduced dentate neurogenesis has been associated with several animal models of neurological conditions (e.g. Alzheimer disease, temporal lobe epilepsy or depression), but there isn’t clear evidence whether these changes are a cause or result of the pathologic symptoms (Zhao et al., 2008; Sloviter et al., 2010; Aimone et al., 2011; Srikumar et al., 2011). Dentate neurogenesis is closely related to performance on some hippocampus-dependent behavioral tasks, such as pattern separation in the DG (Sahay et al., 2011), memory consolidation or anxiety-related behaviors (Riedel et al., 1999; Broadbent et al., 2004). The embryonic origin of the adult neural stem cells (NSC) of the DG is the ventricular zone of the ventral hippocampus. These cells originate in the last prenatal week and migrate to and settle in the DG during the two first postnatal weeks (Li et al., 2013). These adult NSCs are responsive to the morphogenic signaling molecule Sonic Hedgehog (Shh) and consequently express Gli1 (Ahn et al., 2004; Ahn and Joyner, 2005). Shh is important for the establishment and maintenance of the stem cells in neurogenic niches in the adult brain (Machold et al., 2003; Ahn and Joyner, 2005; Breunig et al., 2008; Han et al., 2008). Shh is also involved in the origin of these cells during development from the ventral hippocampus and during their transit to the DG where they become part of the NSC population (Li et al., 2013). Consistent with this, experiments using fate mapping with transgenic mice revealed that embryonic dentate NSCs are responsive to SHH during late gestation before they settle in their permanent niche in the SGZ (Ahn & Joyner 2005; Li et al. 2013). These cells maintain their responsiveness while quiescent during adulthood (Ahn & Joyner 2005; Encinas et al. 2011). Although, we already know several features about the development of the SGZ, there are multiple questions that still remain unclear. How Shh regulates these events or what other molecules may coordinate with Shh in DG development are largely unknown.
The transcription factor Emx2 is expressed specifically in the hippocampus, starting in early development. This expression is maintained in cell progenitors and specifically in the SGZ during the second postnatal week (Mallamaci et al., 1998). Prominent defects in the hippocampus and dentate gyrus of the Emx2-/- mutant mice have been well described in previous studies (Pellegrini et al., 1996a; Oldekamp et al., 2004; Zhao et al., 2006). For instance it is known that the shape and size of the hippocampus is modified and reduced and the DG is almost missing in these mutants (Simeone et al., 1992; Pellegrini et al., 1996b; Tole et al., 2000; Oldekamp et al., 2004). Closer analyses of the DG showed a lost of GC and disorganization of the radial glial fibers used as scaffold by the putative granular cells to reach their final destination (Rickmann et al., 1987; Oldekamp et al., 2004)
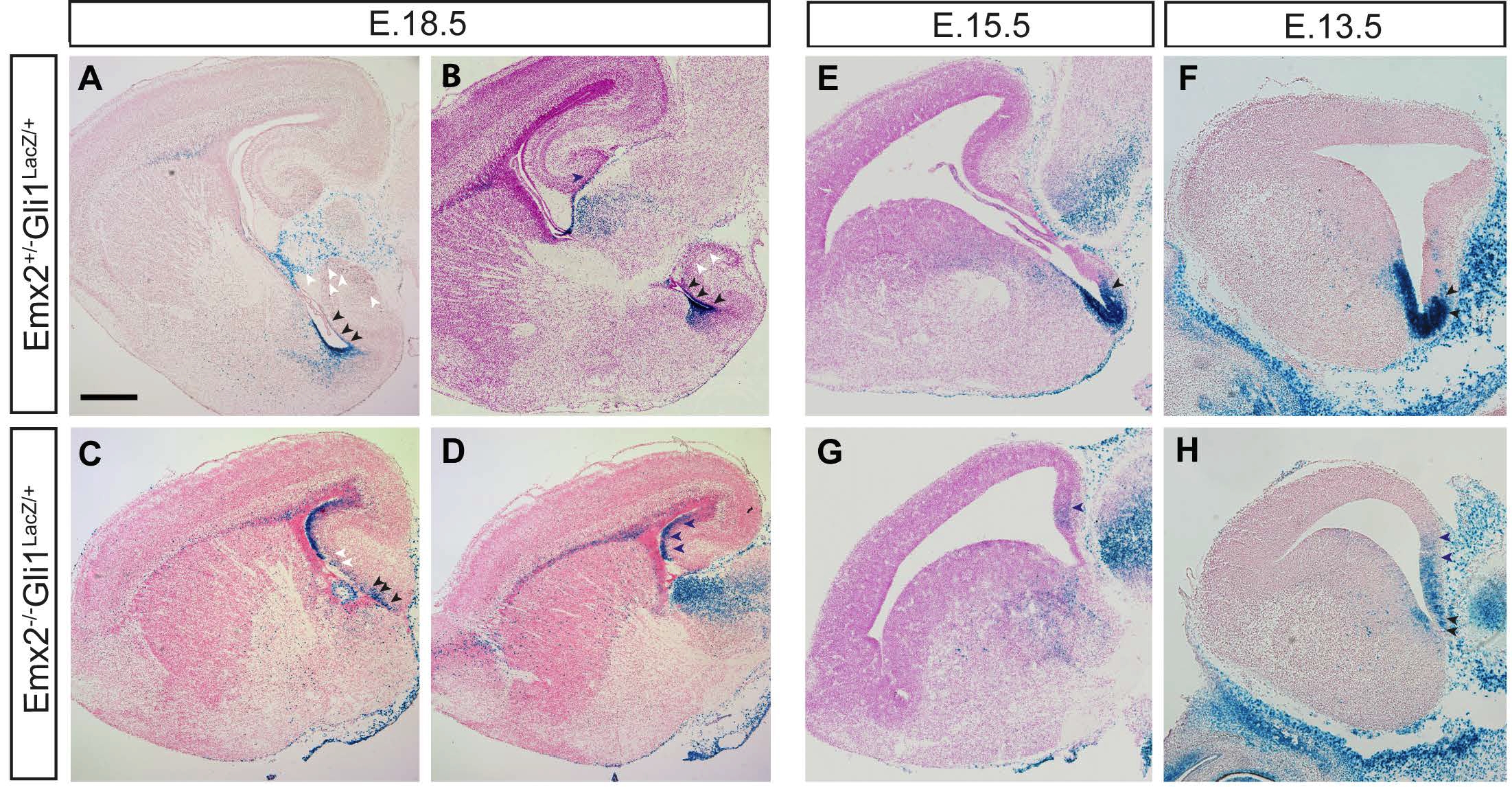
Figure 1. Static distribution of Shh responsive cells in mice lacking Emx2 gene show an ectopic distribution in dorsal ventricular zone (dVZ) of most lateral sections of hippocampus. Sagittal sections of Emx2+/-Gli1LacZ/+ at E18.5 (A and B), E15.5 (E) and E13.5 (F) and Emx2-/-Gli1LacZ/+ mutant mice at E18.5, (C and D); E15.5, (G) and E13.5, (H) showing the expression pattern of LacZ protein under the Gli1 promoter in lateral levels of the hippocampus (dorsal and ventral regions). ).Scale bar (in A) A-H, 500μm.
The coincident expression pattern of Shh and Emx2 in the amygdalo-hippocampal region (see Brain Allen Atlas) and the dramatic defects that Emx2 mutant mice have in the DG directed us to study the embryonic DG in Emx2 mutant mice to the role of Emx2 in the origin and development of the SGZ or more precisely, the cells that will become adult NSC.
Here, we analyzed the population of Gli1+ cells in the Emx2 mutant mice to find to further elucidate the molecular step directing migration of embryonic NSC from the ventricular zone of the ventral hippocampus to the SGZ of the dorsal region. We find that Emx2 is involved in the Shh pathway not only during the migratory stream to reach the dorsal region but also in the proliferative region where the embryonic NSCs originate. The loss of Emx2 causes a reduction in Gli1+ cells that would be part of the population of the adult NSCs in the ventral VZ and some of Gli1+ are relocated in ectopic regions at dorsal VZ where they also accumulate markers of proliferative cells.
Methods and Materials
Animals
The following mouse lines were obtained from Jackson Laboratory (Bar Harbor, ME): Gli1- nLacZ (stock#008211), Gli1-CreERT2 (stock#007913), Ai14 (stock#007914), Shh-gfpcre (stock#005622), Emx2 mutant was kindly provided by Dr. John Rubenstein (UCSF). The day of vaginal plug was considered embryonic day 0.5 (E0.5). Mouse colonies were maintained at UCSF in accordance with National Institutes of Health and UCSF guidelines. Embryos were collected via cesarean section, fixed in 4% paraformaldehyde for 2 hours (E15.5) or perfused and then post-fixed 2 hours (E.18.5), processed through a sucrose gradient, and embedded in OCT for cryosectioning. Embryos were cryosectioned at 16 μm. All animal protocols were approved by the University of California, San Francisco Institutional Animal Care and Use Committee.
LacZ staining
X-gal staining was developed at 37°C overnight in the staining solution [5mM K3Fe(CN)6, 5mM K4Fe(CN)6, 5mM EGTA, 0.01% deoxycholate, 0.02% NP40, 2mM MgC12, 1mg/ml X-gal]. Sections were post-fixed with 10% formalin at room temperature overnight. Slides were then counterstained with nuclear-fast red (H-3403, Vector Laboratories) at room temperature for 10min before proceeding for dehydration (70%, 95%, 100% ethanol, xylene twice) and coverslipping with Mount-Quick (Ted Pella).
Tamoxifen induction
Tamoxifen (T5648, Sigma) was dissolved in corn oil (C8267, Sigma) at 20mg/ml. Pregnant females were administrated intraperitoneally (I.P.) with 2mg of tamoxifen per 40g animal. (BrdU)?
Immunohistochemistry
Immunohistochemistry was performed as previously described [10], [23] using the following antibodies: mouse anti-BrdU (1:75, BD Biosciences); rat anti-Ctip2 (1:500, Abcam); rabbit anti-Ki67 (1:400, Thermo Scientific); rabbit anti-Pax6 (1:500, Abcam); rabbit anti-Tbr2 (1:1000,Abcam); chicken anti-GFP (1:500, Aves lab); rabbit anti-Prox1 (1:1000); rabbit anti-Blbp (1:500,Chemicon); rat anti-GFAP (1/200, Thermofisher); rabbit anti-GFAP (Dako, 1/500) mouse anti-nestin (Chemicon, 1/500); rabbit anti-phospho-histone 3 (Upstate, 1/500) rabbit anti-DsRed (Clontech, 1/200); rabbit anti-Olig2 ()? BrdU?. Primary antibodies were detected using secondary antibodies conjugated to Alexa fluorophores (Invitrogen). Stained sections were visualized on a Nikon fluorescent microscope and captured with a digital CCD-cooled camera and QCapture Pro software (QImaging Surrey). Composite images were prepared in Adobe Photoshop CS5 and Adobe Illustrator CS5. Contrast, color and brightness were adjusted in Adobe Photoshop CS5.
Statistics
All statistical analyses were done on SPSS15 software. The statistical significance of single comparison on discrete data was performed using the nonparametric Chi-square’s test. For continuous data the statistical significance of single comparisons was performed using two-tailed ttest with Welch’s correction when required (non-equal variances) or Mann-Whitney nonparametric test when data didn’t fit to a normal distribution (assed by Shapiro-Wilk normality test). For multiple comparisons we used ANOVA with a Tukey HSD posthoc to determine the significance between groups after checking our data fitted to a normal distribution (assed by Shapiro-Wilk normality test) and the variance was equal by Levene’s test.
Results
Ectopic distribution of Hh responsive cells in the hippocampus of Emx2-/- mutant mice To examine the idea that Emx2 and Shh expression are coordinated we first studied the localization of Gli1 positive cells during development in the hippocampus of the Emx2-/- mutant mice by crossing the mutants with Gli1nLacZ transgenic reporter mice (which express LacZ under the Gli1 promoter) (Fig 1, A-H). This showed that Gli1 positive cells are restricted in the wild type animals to the DG of the dorsal hippocampus at E18.5 (Fig 1, A, B). At similar anatomic levels in the Emx2-/- mutant mice, we found Gli1+ cells neither in the presumptive DG nor in the transitional hippocampus. We also found high expression of LacZ in the VZ/SVZ in the ventral hippocampus of control mice, where these cells are born and start their migration to the DG. In mutants in the same area, this expression is reduced suggesting a defect in the origin and migration of this population of cells (Fig 1, A-D). These findings were also seen at early developmental stages (data not shown). We also found an ectopic population of cells expressing Gli1 in the VZ of the dorsal hippocampus (Fig 1, C-D) at E18.5 in the Emx2-/- mutant mice. We examined the origin of this ectopic population by analyzing Gli1 expression patterns at earlier stages. We found increased Gli1 expression in dorsal regions of the hippocampus at E15.5 (Fig 1, E and G) and E13.5 (Fig 1, F and H) in the Emx2 KO mice corresponding to the VZ of the dorsal hippocampus at later stages (Fig 1, C-D). Since previous studies have shown that Shh has an important role in the origin and maintenance of the LLNSC of the SGZ in the DG and that Gli1+ cells are responding to Shh, and are NSC (Ahn and Joyner, 2005), we wished to further study and characterize the misexpression of Gli1+ cells along the dorsal prospective hippocampus and the reduction observed in the ventral ventricular zone of the hippocampus. To do this we used the Gli1CreERT2 conditional transgenic mice crossed with Ai14F/F reporter line as a tool to follow and characterize the Gli1 positive cells and to determine how the lack of Emx2 affects the population of NSC originating embryonically in the ventral region of the hippocampus.
Fate mapping of Shh responsive cells in the Emx2-/- mutant mice
Emx2-/- mutant mice died at birth due to kidney problems (Simeone et al., 1992), so our study includes stages up to late gestation when embryonic stem cells are born in the ventricular zone of the ventral hippocampus (Li et al., 2013). To better understand how Hh responsive cells are distributed in the Emx2-/- mutant mice, we analyzed the fate map of Gli1+ cells using Gli1CreERT2; Ai14F/+ mice bred into the Emx2-/- line. E17.5 embryos were injected with tamoxifen and then analyzed one day later (Fig 2, A-D). In the dorsal hippocampus of control mice we found that a small group of recombined cells had already settled in the DG at lateral levels (Fig 2, A). Also, some sparsely distributed cells were found in the dorsal ventricular zone and at the meninges. On the other hand, the Emx2-/- mutant did not have labeled cells in the presumptive DG and there was an accumulation of recombined cells in the dorsal ventricular zone and at the meninges (Fig. 2, B). In the ventral ventricular zone of the hippocampus we found a reduction of AI14+ cells in the Gli1CreERT2 mutant mice compared to the controls (Fig. 2, C-D). We also found that the phenotype found in the Gli1CreERT2 mice was similar to that seen in the Gli1nLacZ mice both in the dorsal and ventral hippocampus. This similarity in the expression pattern between the static distribution of Gli1+ cells obtained with the Gli1nLacZ and the short term fate mapping allowed us to characterize the phenotype of the Gli1+ ectopic cells found in the dorsal ventricular zone (Fig 3) and the changes found in the ventricular zone (Fig 5) of the ventral hippocampus.

Figure 2. Loss of Emx2 leads to an accumulation and reduction of Gli1+ cells in the ventricular zone of dorsal and ventral hippocampus respectively. Sagittal sections through the hippocampal region of Emx2+/-Gli1CreERT2+/-Ai14F/+ (control) (A,C) and Emx2-/-Gli1CreERT2+/-Ai14F/+ (mutant) (B,D) E18 mice after E17.5 tamoxifen injection showing the distribution of AI14+ (red) cells after the immunohistochemistry and co-stained with DAPI in dorsal (A-B) and ventral (C-D) hippocampus.(CA1, cornus ammonis 1; CA3 cornus ammonis 3; DG, dentate gyrus; F, fimbria; VHip, ventral hippocampus; dVZ, dorsal ventricular zone; vVZ, ventral ventricular zone). Scale bar (in A) A-D, 50 μm.
The ectopic population of Gli1+ cells in the hippocampal dVZ are progenitors
Previous studies of Emx2-/- mutant mice showed a lack of GC in the DG (Pellegrini et al., 1996; Yoshida et al., 1997) and although the mutant dentate is genetically well specified, it does not grow and develop properly (Tole et al., 2000). We have shown in the Gli1nLacZ and the Gli1CreERT2 mutant mice expanded expression of Gli1+ cells in the VZ of the dorsal hippocampus at lateral levels (Fig 1). In order to understand what these Gli1+ cells are we first characterized them with multiple markers.
To assess whether some of the Gli1+ cells belong to the glial lineage we study the coexpression of GFAP (Fig 3, A and B), and Blbp (Fig 3, C and D) with Td-tomato by immunohistochemistry in controls and Emx2-/-Gli1CreERT2; Ai14F/+ mutant mice. GFAP and Blbp are also expressed in neural progenitor cells at embryonic stages (Giachino et al., 2014; Seki et al., 2014) and GFAP labels the fibers of the radial glial cells. We found almost no co-expression of GFAP with AI14+ cells in control animals at E18.5. Only a very few GFAP+ cells appeared around the pia and close to the meninges (Fig 3, A, arrow). In Emx2-/- mutant mice, on the contrary, we saw that some of the Ai14 recombinant cells expressed GFAP at the dorsal border of the fimbria (Fig 3, B, arrow). We found that control mice had only a few Ai14+ cells expressing Blbp in the Fimbrio-Dentate Junction (FDJ) (Fig 3, C, arrow) and none in the dVZ, however, in Emx2-/- mutant mice, several of the ectopic recombinant cells in the dVZ expressed Blbp (Fig 3, D, arrow).

Figure 3. The ectopic population of Shh responsive cells in Emx2-/- mutant mice colocalized with diverse progenitor cell markers. Sagittal sections of dorsal hippocampus of Emx2+/-Gli1CreERT2+/- Ai14F/+ control, (A, C, E, G, I and K) and Emx2-/-Gli1CreERT2+/-Ai14F/+ mutant E18.5 mice (B, D, F, H, J and L) after received a tamoxifen injection at E17.5 and showing the coexpression of Ai14 with GFAP (A and B), Blbp (C and D), Nestin (E and F), BrdU (G and H), Sox2 (I and J) and Tbr2 (green) (K and L) immunohistochemistry co-stained with DAPI to delimit the areas. (CA1, cornus ammonis 1, CA3 cornus ammonis 3, DG, dentate gyrus, F, fimbria, dVZ, dorsal ventricular zone of the hippocampus; arrowhead, recombinant cells expressing Ai14 (red); arrow, cells colocalizing Ai14 (red) with one of the analyzed maker (green)). Scale bar (in A) A-L, 50 μm.
Taking advantage of GFAP labeling of the radial fibers used as scaffolding for the newly born neurons to reach the DG (Eckenhoff and Rakic, 1984; Rickmann et al., 1987), we next checked the shape of the dentate scaffolding. In control mice, GFAP is strongly expressed at the border of the fimbria and fibers extend to the entrance of the hilus and project to the pia around the forming dentate (Fig 3, A). In the Emx2-/- mutant mice, fibers are also seen at the border of the fimbria but don’t follow the pial surface and instead spread out in a disorganized way to reach the presumptive dentate (Fig 3, B), probably affecting the radial migration of the granule cells. We also checked the oligodendrocyte lineage, labeling the cells with Olig2, but we found no expression in the dVZ of the hippocampus at this age both in control or mutant mice (Supplemental Fig 2, A, B).
To confirm our impression that the ectopic Gli1+ cells correspond to progenitor cells, we next studied the colocalization of several progenitor markers such as Nestin, Sox2 and Tbr2. As with the GFAP and Blbp we found multiple Nestin+/Ai14+, Sox2+/AI14+ and Tbr2+/AI14+ cells in the dVZ of the hippocampus in Emx2-/- mutant mice and none in the controls (Fig 3, E, F, G-L). Therefore we conclude that the loss of the Emx2 gene leads to a misexpression of Gli1 in progenitor cells of the dorsal hippocampus. We also examined whether these Gli1+ cells were proliferating with acute BrdU labeling (Fig 3, G and H). Although the distribution of BrdU in the VZ and SVZ of the dorsal hippocampus seems similar in controls and Emx2-/- mutant mice, the Ai14+ ectopic cells localized in the dVZ of the hippocampus were also stained with BrdU (arrows in Fig 3, H), pointing out that most of the Gli1+ cells in the dVZ are dividing.

Figure 4. Emx2 mutant mice show an increase in the density of PH3 and Tbr2 positive cells in the VZ of the dorsal hippocampus. Sagittal sections of E18.5 (A-C and E-G) wt (A, E), heterocygous (B, F) and Emx2 mutant mice (C, G), showing immunohistochemistry of PH3 (A-C), or Tbr2 (E-G) marker. Quantification (D, H) of the expression of PH3 (D) or Tbr2 (H) in controls (red bar) het (white bars) and Emx2-/- mutant mice (black bars) by cell density in the VZ of dorsal hippocampus. (CA1, Cornus Ammonis 1, CA3 Cornus Ammonis 3, DG, dentate gyrus, F, fimbria, dVZ, dorsal ventricular zone). Scale bar (in A) A-C, E-C, 50 μm.
Increased number of proliferating and secondary progenitor cells in the dVZ in Emx2-/- mutant mice
Previous studies have shown the loss of Emx2 affects the proliferation of progenitor cells in the hippocampus (Muzio et al., 2005). Our experiments show that most of the progenitors expressing Gli1 in the Emx2 mutant mice are in a proliferative state (Fig 3, C, J). To understand better if the loss of Emx2 affects the cell cycle these progenitors, we carried out experiments to quantify the density of those dividing cells in the dVZ of Emx2 KO and controls at E18.5. The analysis of PH3 in Emx2+/+, Emx2+/- and Emx2-/- mutant mice showed an approximate doubling of cell density of these proliferative markers in Emx2-/- mutant mice (Fig 4, A, B, C and D). We found no statistical differences between Emx2+/- and Emx2+/+ in PH3 and Ki67. We next analyzed the intermediate progenitors using Tbr2 as a marker in the dVZ of the hippocampus. We found an increase of ~30% in the density of Tbr2+ cells in the in Emx2-/- compared with Emx2+/- and Emx2+/+ mice (Fig 4, E-H). Since ectopic expression of Gli1 in the dorsal hippocampus starts as early as E13-E15 (Fig 1, E-H) we then analyzed the expression of PH3 and Tbr2 at E15.5 and found an increase of ~60% of PH3+ cells (Fig 5, A-D) in the Emx2-/- compared with Emx2+/- and Emx2+/+ mice. On the other hand we did not find any difference between the three groups of animals in Tbr2+ cells at this stage (Fig 5, E-H). These results further suggest that the ectopic population of Gli1+ located in the dVZ of the hippocampus correspond to progenitor cells.
When we examined E18.5 embryos we observed differences in the distribution of PH3+ and Tbr2+ cells in the mutants compared with controls. PH3+ cells in Emx2-/- mutant mice at E18.5 appeared mainly in the VZ of the dorsal hippocampus, just at the border of the ventricle, while in controls the population of PH3+ cells was homogeneously distributed in the whole hippocampus, including FDJ and DG itself, suggesting that in the mutant, the population that should be dividing in the FDJ and DG at that stage is not there or is not able to proliferate. The intermediate progenitor marker Tbr2 were differentially distributed in Emx2-/- and Emx2-/+ or Emx2+/+ mice as well, with its distribution in the Emx2-/- mutant mice restricted to the dVZ with a very few cells close to the fimbria, in the FDJ or the DG compared to control mice. Together, the E18.5 and E15.5 data suggest a role for Emx2 through Gli1 in the control of the cell cycle length, perhaps by lengthening the cell cycle as other authors have proposed (Muzio et al., 2005). The increase of Tbr2 density number at E18.5 and not earlier at E15.5 in the Emx2 mutant mice could be a result of a temporal change in the expression pattern or an accumulation of the cells due to a migration problem, perhaps because the glial scaffolding is not well formed (Fig 4 A, D). To explore the second possibility we measured the VZ of the dorsal hippocampus. The analysis of the VZ width displayed a ~4-fold increase in the Emx2 mutant mice compared to controls (Fig 6, A). We next wondered whether the increase in the proliferation rate in the VZ in the Emx2-/- leads to an increase in the number of immature neurons in that region. So we next performed a X-gal staining in the mice line Emx2;NeuroDLacZ in which lacZ is expressed under the promoter NeuroD, a postmitotic immature neuronal marker. The analysis of LacZ expression showed an accumulation of immature neurons in the VZ/SVZ zone of the Emx2 mutant compared with controls (Fig 6, D, E).

Figure 5. Loss of Emx2 gene leads to an increase in the dorsal hippocampus proliferation at E15.5. Sagittal sections of E15.5 dorsal hippocampus in Emx2+/+Gli1CreERT2+/-Ai14F/+ (A and E); Emx2+/-Gli1CreERT2+/-Ai14F/+, (B and F) and Emx2-/-Gli1CreERT2+/-Ai14F/+ mice (C and G), showing distribution of Phospho-Histone 3 (PH3) (A, B and C) and Tbr2 (E, F and G) markers after immunohistochemistry. Quantification (D, H) of the expression of PH3 (D) or Tbr2 (H) in controls (red bar) heterozygous (white bars) and Emx2-/- mutant mice (black bars) by cell density in the VZ of dorsal hippocampus. (Hip, hippocampus, dVZ, dorsal ventricular zone). Scale bar (in A) A-C, EC, 50μm.
Dorsal hippocampal Shh expression is minimally changed in Emx2-/- mutant mice
The misexpression of Gli1 in the Emx2-/- mutant mice in progenitor cells pointed us to a possible change in Shh expression. Detection of Shh by immunostaining or in-situ hybridization (ISH) in the brain is very difficult due to low level expression, therefore we chose to use the Cre transgenic line under control of the Shh promoter, ShhGFPCre, crossed with the Ai14Flox reporter line to follow the Cre positive cells which are red because of tdTomato. In this way we could examine the numbers and distribution of cells generated from the Shh expressing lineage and thus get a sense of whether the Shh expressing domains in the brain are altered. Using this approach we found no clear differences in the distribution and numbers of cells generated from Shh expressing cells at E18.5 (Fig 7, A-D) (Fig 7, arrowheads in B and D). However, as the ShhGFPCre also has GFP expression under the Shh promoter, we can also follow the cells that are actually expressing the protein at this age. We did not find any expression of GFP in cells close to the dVZ so we are unable to identify the specific source of Shh that might affect the ectopic distribution of Gli1+ cells. These data imply that rather than a change in the distribution of Shh expression leading to ectopic Gli1 that instead the loss of Emx2 may change Shh responsiveness in some way.

Figure 6. Loss of Emx2 leads to an increase of the VZ and the immature neuronal marker NeuroD. Sagittal sections of the VZ of the dorsal hippocampal region showing DAPI (A, B) and LacZ (D, E) staining at E18.5 of controls -(A, D) and mutant (B, E) mutant mice. D and E sections were counterstain with nuclear fast red to visualized anatomy (C) Quantification of the VZ thickness in controls (white bar) and Emx2-/- mutant mice (black bar). Scale bar (in A) A, B 25 μm; Scale bar (in D) D, E, 50 μm.
The Emx2-/- mutant mice have defects affecting the ventral source of NSC
Next we studied the role of Emx2 gene in the VZ of the ventral hippocampus (vVZ). The histological analysis of the vVZ and the expression pattern of Gli1 revealed a reduction in the number of Gli1+ cells as well as the size of the ventricle at E13.5, E15.5 and E18.5 in the Emx2-/- mutant mice (Fig 1). To pinpoint what kind of cells were affected we performed a broad characterization of the cells in this region. Immunohistochemistry performed at E18.5 of glial markers as GFAP, Blbp, or progenitors as Nestin and Sox2 showed a reduction in their expression in the Emx2-/- mutant mice compared to controls (Fig 8, A-F, I, J). We also observed a reduction in the number of intermediate progenitors, labeled by Tbr2, and the general mitotic marker BrdU. Hence we concluded the loss of Emx2 leads to a reduction of the number of progenitor cells and cell division in the vVZ. The Gli1+ cells generated in this region during the last week of gestation migrate from the caudal (temporal pole) to the dorsal (septal pole) to populate the DG and be part of the SGZ and become the long-lived neural stem cells (LL-NSC) (Li et al 2013). We wished to investigate whether the reduction of progenitor cells and the divisions in the vVZ in the Emx2-/- mutant mice could be also affecting to the population of LL-NSC. As explained before, we cannot follow Emx2-/- mutant mice after birth, but we were able to describe defects when they first appear at E17.5, and follow them to E18.5 (Fig 2, A-E). We performed fate-mapping experiments by injecting tamoxifen at E17.5 and analyzing one day later the number and location of the cells originating from Gli1+ cells in the Emx2 transgenic mice. E18.5 Emx2-/-;Gli1CreER-2 mutant mice showed a reduction of Ai14 recombinant cells in the vVZ compared to controls. We also explored the migratory pathway of the Gli1+ cells (Fig 2, C, D) born in the vVZ and found Ai14+ cells en route to the DG in controls (Fig 2, C), whereas in the Emx2-/- mutant mice we found no cells in this migratory route (Fig 2, D). These data provide evidence that Emx2 is required for the generation and migration of the Gli1+ cells.
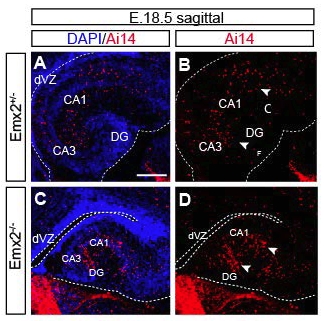
Figure 7. The expression of Shh in the hippocampus. Sagittal sections through the dorsal hippocampus of E18.5 (A-D) Emx2-/+;ShhCREGFP;Ai14F/+ (A, B) and Emx2-/- ;ShhCREGFP;Ai14F/+ (C, D) embryos showing the expression of Ai14 under Shh promoter. (CA1, Cornus Ammonis 1, CA3 Cornus Ammonis 3, DG, dentate gyrus, dVZ, dorsal ventricular zone). Scale bar (in A) A-D, 100 μm.
Loss of Emx2 leads to reduction of Shh+ cell density in the amygdalo-hippocampal region
Previous data have suggested that Shh is required for the origin and maintenance of the LLNSC (Han et al., 2008; Ihrie et al., 2011; Li et al., 2013). In order to understand if the reduction in the number of Gl1+ progenitor cells in the ventral VZ was regulated directly by the Emx2 gene or indirectly through Shh we then next analyzed the expression pattern of Shh in the Emx2 mutant mice. To perform the analysis we used again the Emx2;ShhCreGFP crossed with the AI14 reporter line where GFP expression shows the present activity of Shh and the Tdtomato the activity at any time (present or past). We found a reduction by half of the density of GFP+ and Tdtomato+ cells in the amydalohippocampal region of the Emx2-/-; ShhCreGFP mutant mice at E18.5 compared to controls (Fig 9), this was associated with apoptosis at the same age in the same region (Fig 8). Hence we conclude that Emx2 is crucial for the survival of the Shh+ cells in the amydalo-hippocampal region and most likely reduction of Shh protein contributes in the origin and maintenance failure of the Gli1+ cells.
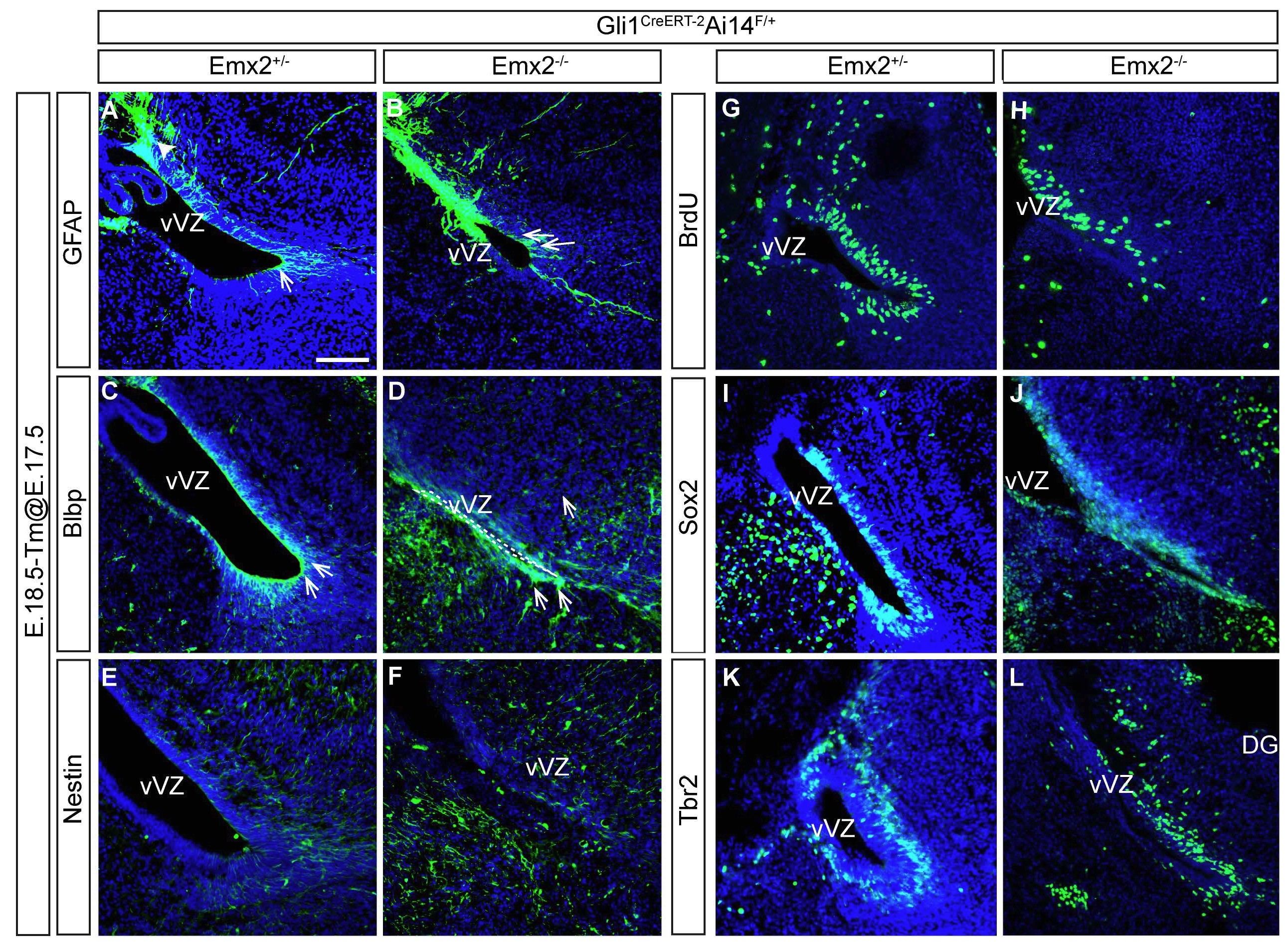
Figure 8. The vVZ of the Emx2-/- mutant mice is smaller and the ventricle is thinner than the wt mice. Sagittal sections of ventral hippocampus in Emx2+/- control mice, (A, B, C, G, H and I) and Emx2-/- mutant mice (D, E, F, J, K and L) at E18.5 showing the expression of diverse markers in vVZ with GFAP (A and D), Blbp (B and E), Nestin (C and F), BrdU (G and J), Sox2 (H and K) and Tbr2 (I and L). (CA1, Cornus Ammonis 1, CA3 Cornus Ammonis 3, DG, dentate gyrus, F, fimbria, dVZ, dorsal ventricular zone of the hippocampus; arrowhead, recombinant cells expressing Ai14; arrow, cells colocalizing Ai14 with one of the analyzed maker). Scale bar (in A) A-L, 50μm.
Discussion
The lack of Emx2 alters the population of the cells responding to Shh in the hippocampus
The identification of the origin of the LL-NSC in the ventricular zone of the ventral hippocampus, their posterior migration through the longitudinal axis and the distinct source of Shh involved in the formation of the SGZ, are important observations allowing us to better understand hippocampal and SGZ development. The analysis of the expression pattern of cells that respond to Shh is a powerful tool to elucidate the embryonic origin of the cells that will became the NSCs in the adult SGZ (Ahn & Joyner 2005). These cells, unlike SVZ NSCs, are born embryologically in a different region from where they will settle in the adult (Altman & S. a Bayer 1990). One of the many questions remaining is to identify the molecular mechanisms involved in the origin and migration of the LL-NSC. The analysis of genes expressed in the ventral hippocampus and in the amygdalo-hippocampal region directed us to study the Emx2-/- mutant mice as a tool to understand the development of the LL-NSC.
As we previously showed, the analysis of Shh responding and expressing cells with the transgenic mice Gli1nlacZ and Gli1CRE-ERT2 and ShhCreGFP+ facilitates the identification of the LL-NSCs during their development in the hippocampus (Li et al 2013). Loss of Emx2 produces prominent defects in the hippocampus and DG and consequently in the SGZ where the LL-NSCs will settle. In this context, the expression pattern of Shh responding cells appeared altered. LacZ+ cells in the Emx2-/- ;Gli1nLacZ mice at E18.5 compared to the control revealed differences not only in the dorsal hippocampus, where LacZ+ cells in controls first appear at E17.5 (Li et al. 2013), but also in the ventral hippocampus, where the cells that will originate the LL-NSCs and will later populate the SGZ are drastically decreased in number. The defects found in mice without Emx2 are already well described (Tole et al. 2000; Shinozaki et al. 2004; Gangemi et al. 2006; Zhao, Kraemer, Oldekamp, Cankaya, et al. 2006). The shape and size of the hippocampus is modified and reduced and the DG is almost missing in these mutants (Oldekamp et al. 2004; Pellegrini et al. 1996b; Tole et al. 2000; Simeone et al. 1992). Our analysis of Gli1 expression showed no Gli1+ cells in the hippocampal plate at E18.5, in the DG or in the transitional hippocampus where Gli1+ cells are already present in control mice. Also it is well known that Emx2-/- mutant mice showed a loss of GC, described as a diminished in the number or indeed total lack of Prox1+ cells (Oldekamp et al. 2004), that is in agreement with the lack of Gli1+ cells en route to the dorsal dentate.
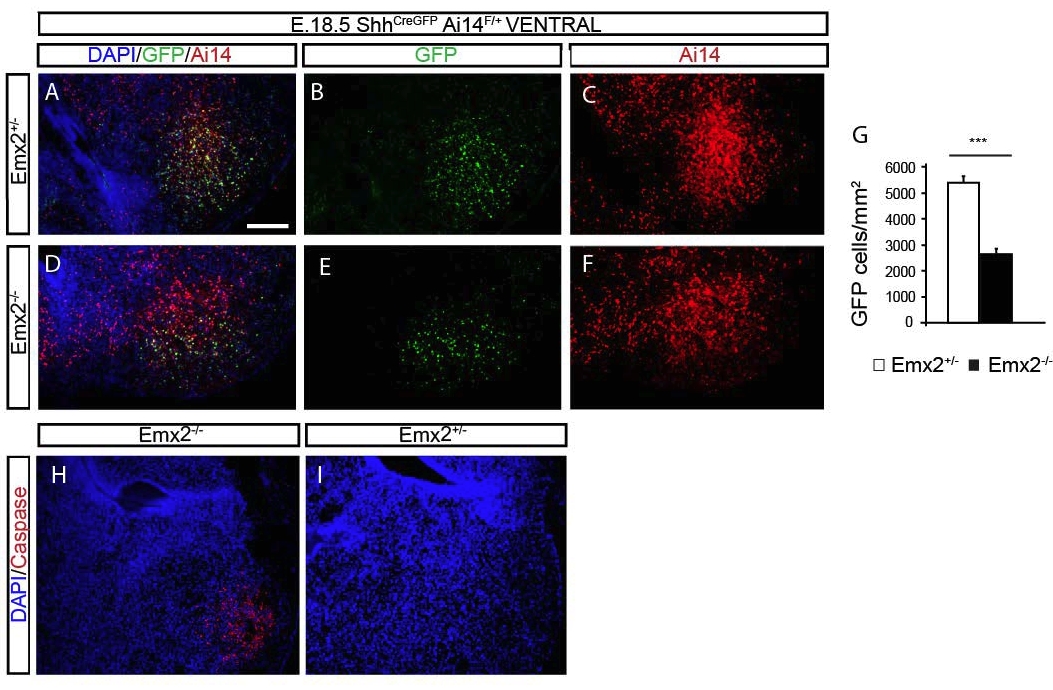
Figure 9. Loss of Emx2 lead to a reduction of the Shh+ cells in the amygdalo-hippocampal region. Sagittal sections through the amygdalo-hippocampal region showing the expression of Ai14 (A, C, D, F), GFP (A, B, D, E), DAPI (A, D) and caspase in Emx2-/+;ShhCREGFP;Ai14F/+ (A-C) and Emx2-/-;ShhCREGFP;Ai14F/+ (C-F) mutant mice at E18.5 (A-F) Scale bar (in A) A-D, H, I 100μm.
The ventricular zone of the ventral hippocampus is reduced in the Emx2-/- mutant mice
Cells originating in the VZ of the ventral hippocampus become the LL-NSC that will populate the SGZ. This population of cells responds to a source of Shh located in the amygdala and has to migrate to reach the SGZ in both the ventral and dorsal DG (Li et al 2013). The VZ of the ventral hippocampus is smaller in the Emx2-/- mutants suggesting that the population of embryonic NSC could be affected, and this was supported by our analysis of progenitor markers and cell division.
Since it has been proved that Shh plays a role in the development of the SGZ (Breunig et al. 2008; Han et al. 2008; Machold et al. 2003) and most likely the source of Shh in the amygdala is the one involved in the origin and maintenance during migration of the LL-NSC from the VZ of the ventral hippocampus (Li et al. 2013), we checked the status of the Shh producing cells in the amygdala. The results showed a reduction in the density of GFP+ cells in the ShhCreGFP+;Emx2-/- transgenic mice and an increase of Caspase+ cells in the amygdala region. Our results showed Emx2 gene is necessary for the survival of the Shh-producing cells in the amygdala.











