Malignant gliomas are primary brain tumors considered to be one of the deadliest cancers. Despite surgical intervention followed by aggressive radio- and chemo-therapies, average survival is approximately 15 months of diagnosis. Recurrent tumors resembling all the characteristics of the original tumor mass and growing in close vicinity to the original site are frequent due to presence of a self-renewing population of cells, glioma stem cells. The cells are resistant to therapies and able to invade the surrounding healthy brain tissue. Indeed, infiltrative growth assisted by numerous interactions with microenvironment are hallmarks of glioma growth. Many research efforts are put forward to understand the mechanisms of invasion. Glioma cells adopted numerous biological strategies to their own advantage to viciously propagate and navigate narrow spaces within the brain. Despite enormous amount of data on malignant gliomas generated by –omics approach which broaden our knowledge on glioma physiology in the last decade, parallel success in discovering new therapies did not happen. Thus, new therapeutic approaches may employ healthy cells of the microenvironment to tame malignant growth are necessary. Here, we highlight current knowledge on glioma origin, infiltrative growth, interactions with the microenvironment and potentials for new therapies.
Introduction
Primary brain tumors, PBT are neoplasms originated from various normal brain cells and grow within the brain parenchyma. While non-neuronal glial cells can give rise to several types of PBT, collectively called glioma, the ones originated from astrocytes or their precursors are the most common. Based on histopathological characteristics and criteria established by World Health Organization, WHO, astrocytomas are divided in four (I-IV) grades (Louis et al., 2007). Glioblastoma multiforme, GBM, classified as a Type IV glioma, is the most frequent one with the worst prognosis among them. Though incidence is relatively low, over 22,000 cases per year in USA, GBM remains one of the deadliest cancers. Patient’s perspectives are very grim once they are diagnosed with GBM - median survival time is 15 months despite aggressive therapies such are surgical removal, chemotherapy and radiation. Therapies have been shown ineffective mainly due to reappearance of the more aggressive tumor cells in close proximity to the site of origin, 1-2 cm from the surgical intervention. GBM metastasize to other organs rarely (0.4-2%), which could be accounted for several reasons. One possible cause is due to very short patient survival after initial diagnosis, which does not allow sufficient time for extracranial metastasis to develop. Additionally, it is plausible that blood-brain barrier, BBB poses limitations to potential mechanisms of escape. Finally, there is evidence the immune system plays a role in restricting glioma cells that breached BBB and reached circulation to form secondary tumors (Xie et al., 2014). The later is supported by clinical cases describing patients on immunosuppressive therapy who received transplant organ of an individual with GBM and developed GBM metastasis (Jimsheleishvili et al., 2014). Nonetheless, invading brain parenchyma due to infiltrative nature of glioma growth is well established and considered to be a hallmark of GBM. Many research efforts are put forward to understand the mechanisms of invasion. Indeed, glioma cells adopted numerous biological strategies to their own advantage to viciously propagate and navigate narrow spaces within the brain. Here, we highlight current knowledge on glioma origin, infiltrative growth, interactions with microenvironment and potentials for new therapies.
Glioma Stem Cell
Tumor mass of various cancers has long been described as complex and heterogeneous. This cellular diversity somewhat replicates cellular hierarchy seen in normal tissue organs. Soon after the neural stem cell was described (Uchida et al., 2000), the cancer stem cell, CSC, was first found in an anaplastic astrocytoma (Ignatova et al., 2002), followed by discoveries in medulloblastoma, pilocytic astrocytoma, ependymoma, ganglioglioma (Hemmati et al., 2003; Singh et al., 2004), and GBM (Ignatova et al., 2002; Hemmati et al., 2003; Galli et al., 2004). It is important to emphasize that cancer stem cell does not necessarily form from a transformed normal stem cell. Adult neural stem cells or multipotent neural progenitor cells (Sanai et al., 2005), but also more differentiated lineages within the brain, including neuron-glial antigen 2 cells, NG2 cells (Liu et al., 2011; Sugiarto et al., 2011), astrocytes and even mature neurons (Friedmann-Morvinski et al., 2012) have all been shown to give rise to CSC, albeit the origin of the CSC remains unclear, Figure 1 (for more details see (Lathia et al., 2015)).
Figure 1. Glioma Stem Cells, GSC. Origin of glioma stem cells is still unknown. Tumors are formed of heterogeneous cell population, with only GSCs going through self-renewal.
Glioma stem cells, GSC are generally defined as a self-renewing population of tumor cells with enhanced tumorigenic properties, Figure 1. Nomenclature is still not defined, and various terms including cancer/tumor/glioma/brain tumor stem cell, stem-like tumor cell, cancer-/tumor-/glioma-/brain tumor-initiating cell, and cancer-/tumor-/glioma-/brain tumor-propagating cell are used to describe the cells with common characteristics of self-renewal, giving rise to differentiated progeny, and initiating and propagating tumor growth after transplantation. Here, we are using the term glioma stem cell, the term most frequently used in the glioma field. Due to the abovementioned characteristics, isolating GSC from glioma patient-derived xenograft, PDX models that contain GSC critically advanced our understanding of GBM growth and mechanisms of infiltrative invasion, thus it is considered the best experimental approach available today to study glioblastoma. Unlike traditionally used cultured glioma cell lines propagated in vitro for a long time, GSC in PDX models recapitulate gene expression patterns and invasive intracranial tumor behaviour in mice, similarly to human GBM. Two mouse models able to preserve the characteristics of a patient’s histopathology currently in use are orthotopic transplantation and propagation of the original patient tumor, and heterotypic transplantation of cells into the flank of the nude mouse. The later model, while neing useful due to a lack of interactions with the specific (brain) microenvironment, has limitations. None of the current experimental models completely recapitulates patient pathology, therefore, it is important to acknowledge limitations of the model used in a particular study.
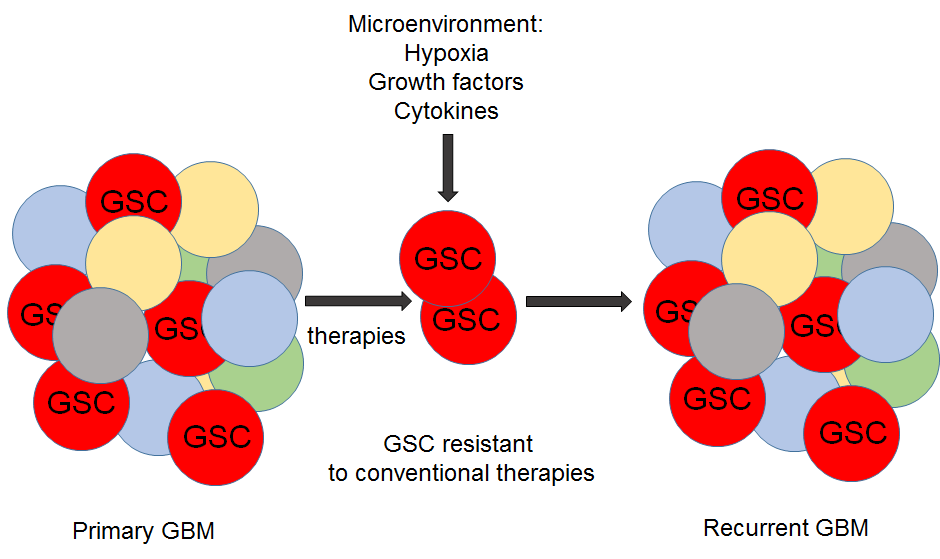
Therapeutic interventions in treating glioblastoma patients are limited and ineffective. After surgical removal of tumor tissue, treatment with radiation therapy together with temozolomide, TMZ, follows. However, GSC has been shown to be resistant to the conventional therapies (Bao et al., 2006; Chen et al., 2012) and to drive GBM recurrence, proliferation, progression and invasiveness, Figure 2. Therefore, defining the cell(s) that initiate glioma growth would allow early and effective treatments by either specifically destroying cells of origin without harming other healthy brain cells or interrupting signaling pathways that allow neoplastic transformation.
Proliferation
The essence of tumor existence is its proliferation, grow and spread. Thus, all biological functions are directed toward that goal. Accumulated mutations, genetic alterations and epigenetic modifications relevant for tumorigenic transformation are mostly geared to the dysregulation of the cell cycle, changes in the tyrosine kinase receptor pathways and the anti-apoptotic retinoblastoma pathway, which altogether allow uncontrollable growth.
Regulation of glioma GSC propagation is governed by several essential mechanisms driven from within the cell and from interactions with surroundings. Main cellular controllers include genetic and epigenetic regulation, and unique metabolic features. Genetic mutations are best documented and extensively studied (Cancer Genome Atlas Research, 2008; Brennan et al., 2013). Besides glioma-specific mutations such are isocitrate-dehydrogenase 1, IDH1 and CIMP (G-CIMP) mutations (Noushmehr et al., 2010), glioma shares similar genetic variations with other neoplasms. Some of them are EGFR, PDGFRA, HDM2, PIK3CA, and TERT promoter and PI3KR1 gain-of function mutations or amplifications and mutations or deletions of the tumor suppressors PTEN, TP53, CDKN2A, NF1, ATRX, and RB1 (for details see (Lathia et al., 2015)). Epigenetic regulation relies mainly on transcriptional factors. Those include MYC which maintains cancer cell survival and proliferation programs (Wang et al., 2008; Zheng et al., 2008; Wurdak et al., 2010; Chan et al., 2012; Fang et al., 2014), STAT3 (Sherry et al., 2009), SOX2 (Gangemi et al., 2009), FOXM1 (Joshi et al., 2013), FOXG1 (Verginelli et al., 2013), GLI1 (Clement et al., 2007), ASCL1 (Rheinbay et al., 2013), ZFX (Fang et al., 2014), NANOG (Zbinden et al., 2010), and ZFHX4 (Chudnovsky et al., 2014). In addition to genetic and epigenetic factors, and consequent abnormalities in signaling pathways, metabolic activity is a strong regulator of GSC proliferation. The glioma stem cell, likewise other tumor cells, has specific metabolic signature. The “Warburg effect” or metabolizing glucose to lactate as a main source of energy, has been described in 1920s by German biochemist and Nobel Laureate Otto H. Warburg. The overproduction of lactate leads to spill of lactate in surroundings, which affects and changes tumor microenvironment perhaps making it more permissive for aggressive dissemination. Non-physiologically high concentrations of lactate and glutamate were measured in patients’ in vivo using microdialysis (Marcus et al., 2010). The shift in metabolism is mainly caused by genetic alterations and preferential expression of set of proteins that support such a shift, for example high affinity glucose transporter, GLUT3, or mutant, IDH-1. Gliomas, similarly to neurons, have a high demand for glutamine, otherwise provided to the brain parenchyma by astrocyte via the glutamate-glutamine cycle (Wise & Thompson, 2010). Preferential interference with mentioned, or some other yet unknown glioma metabolic pathways, may provide a new target for therapies to treat these neoplasms.
Figure 2. Glioma Stem Cells are resistant to therapy. GSCs are resistant to radio- and chemo- therapy. Surviving cells give rise to a new tumor mass if microenviromental conditions are favorable. Hypoxia and various growth factors and cytokines enable GSC to proliferate.
Analysis of GBM sequence database revealed three distinct types of GBM: “proneural” with longer, and “mesenchymal” and “proliferative” with shorter survival probability and more invasive phenotype (Phillips et al., 2006). These findings emphasize the importance of using molecular pathology in profiling GBM as a powerful diagnostic tool in personalized medicine. Comparative gene analysis of proliferative and invading cells indicates differential activation of genes. A distinctive set of proteins is expressed in one or the other behaviour, which was the reason for the introduction of the“go or grow” concept, suggesting that if a cell is in division, it would not migrate, or vice versa, if the cell is moving, it is not going to divide. Cells rather undergo a switching pattern between proliferation and invasion in response to intrinsic and extrinsic factors, cell-cell interactions and the microenvironment. Main drivers of proliferation are upregulation of key pathways Myc, VEGF, bFGF, CSPGs and EGF, combined with external factors of angiogenesis interactions with extracellular matrix, and anaerobic glycolysis. Main drivers of invasion are NF1 loss, CD44, MET, Ras, TGFβ, Zeb1, NFκB, STAT3, c/EBPβ, and miR-451 together with environmental conditions such as hypoxia, ionizing radiation and chemotherapy. However, in-depth analysis suggested that there are some overlapping pathways active in both behaviors such are EGF, TGFβ, NFκB and miR-451 (for more details see (Xie et al., 2014)). Clearly, while the concept is very appealing and mostly correct, it appears a simplified view of glioma behaviour, as they are very complex and heterogeneous cell population.
Infiltrative invasion
The infiltrative growth of single GBM cell invading healthy brain parenchyma is a hallmark of GBM. Identifying key molecules linked to invasion process is critical to creating therapies to control this disease. As indicated above, GSC are a primary source of invasion. The cells are not only resistant to chemotherapy and ionizing radiation, but those conditions prompt them to invasive behaviour (Xie et al., 2014), and later, due to self-renewing properties, are capable of giving rise to a new heterogeneous tumor mass which retains the ability to differentiate into multiple lineages.
As glioma cells move through normal tissue, they face an uphill battle to navigate narrow spaces and degrade the extracellular matrix, ECM mainly composed of proteoglycans, hyaluronan and tenascins (Zimmermann & Dours-Zimmermann, 2008). The cells need extensive adaptation and expression of proteins adept of accomplishing the task. Like their precursors, normal brain cells, neurons and stem cells, glioma cells use similar routes to migrate through the brain. As described by Scherer in the late 1930s (today referred to as Scherer’s structures), glioma cells invade through brain parenchyma, along blood vessels, along with the white matter tracts and in the subarachnoid space underneath the meninges or subpial spread (reviewed in (Cuddapah et al., 2014).
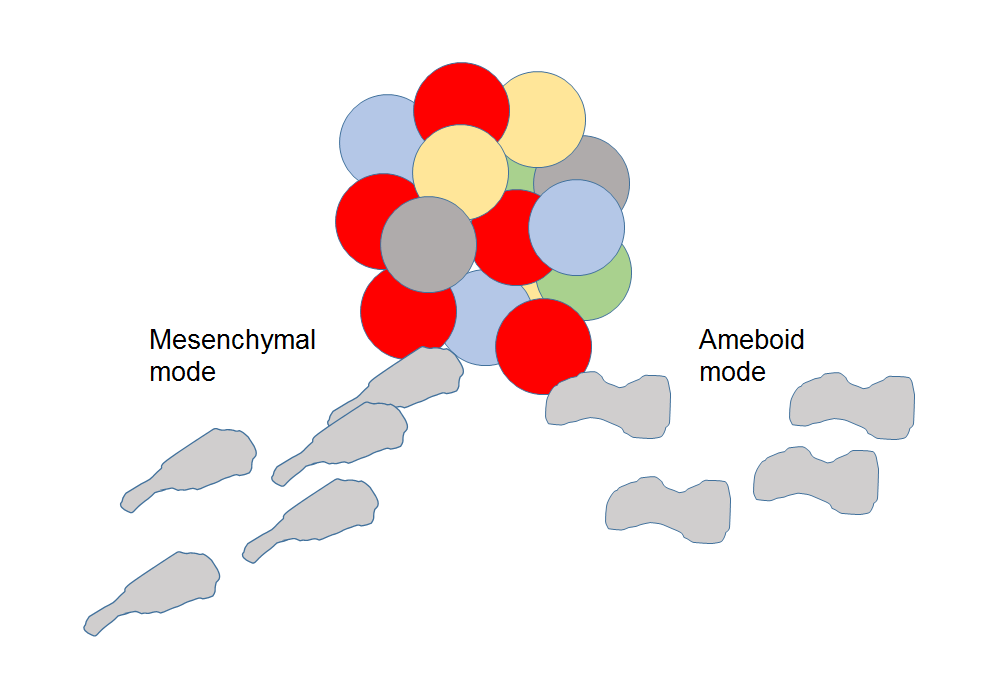
Figure 3. Modes of glioma invasion. Two distinct modes of glioma motility have been described: mesenchymal and ameboid. Mesenchymal invasion is characterized by a train of elongated cells that reorganize ECM and depends on integrin and protease function. As the cells move, they extend specialized proteolytically active plasma membrane protrusions, termed invadopodia, responsible for the focal degradation of the ECM. The process includes complex interactions between the intracellular trafficking, signal transduction and cytoskeleton regulation machineries. Ameboid invasion is a single cell event which utilizes the Rho/ROCK signaling pathway and actin-myosin contractility allowing cells to jam through the ECM.
Cell migration is a complex biological process that requires specific protein machinery, with both intracellular and extracellular components. Actin-myosin molecular motors are responsible for cellular movement (Beadle et al., 2008); cell-cell and cell-ECM interactions through various receptors such are integ¬rins, cadherins and neural cell adhesion molecules, are responsible for attachment, while secretion and activation of matrix metalloproteases, MMPs are responsible for detachment of glioma cells (Kwiatkowska & Symons, 2013).Two distinct modes of glioma motility and interaction with integrins have been described: mesenchymal and ameboid (Vehlow & Cordes, 2013), Figure 3. Mesenchymal invasion is characterized by a train of elongated cells that reorganize ECM and depends on integrin and protease function. As the cells move, they extend specialized proteolytically active plasma membrane protrusions, termed invadopodia, responsible for the focal degradation of the ECM. The process includes complex interactions between the intracellular trafficking, signal transduction and cytoskeleton regulation machineries (Buccione et al., 2009). Ameboid invasion is a single cell event which utilizes the Rho/ROCK signaling pathway and actin-myosin contractility allowing cells to jam through the ECM, Figure 3 (Vehlow & Cordes, 2013).
Irrespective of a mode of invasion and signaling pathways involved, navigating through narrow extracellular spaces (38-64 nm in murine neocortex measured in vivo) puts serious strain on glioma cells as they undergo significant changes in shape and volume. Glioma cells express a number of ion channels activated after the binding of ligands. The channel activation allows movements of water, chloride and potassium ions across the plasma membrane to permit necessary adjustments of shape and size (for details see (Cuddapah et al., 2014)). Interference with functionality of these channels makes them appealing therapeutic targets. Indeed, a specific blocker of Cl− channels, a peptide produced by dessert scorpion (Leiurus quinquestriatus), chlorotoxin, reduces migration of glioma cells (Soroceanu et al., 1999; Lui et al., 2010). Since the toxin binds specifically onto human glioma cells but not to normal brain cells, it has been identified for a potential clinical use (Soroceanu et al., 1998). Chlorotoxin is currently in a Phase I/II clinical trial (NCT00040573) of intracranial injections (Cuddapah et al., 2014).
Microenvironment
The invasion of glioma cells is believed to depend upon crosstalk between various cellular components of the healthy tissue and tumor cells. Thus, it is essential to understand close interactions with healthy brain cells, including astrocytes, oligodendrocytes, neurons, microglia and blood vessels, how they are changing and participating in the invasive process. Recent evidence imply significant impact of microglial and endothelial cells to invasive migration of GBMs. Glioma cells have been shown to produce various autocrine motility factors to enhance invasion (Lyons et al., 2007). Furthermore, the cells express receptors for a range of paracrine factors secreted by normal cells in the brain, like growth factors, neurotrophins, chemokines, cytokines, and kinins, (Hoelzinger et al., 2007). Hypoxic conditions within the tumor initiate expression of various proteins, including some receptors (Zagzag et al., 2008), and changes in normal tissue around it. For example, an increased concentration of bradykinin, BK and formation of its concentration gradient around blood vessels have been associated with hypoxia. Glioma cells respond to BK owing increased expression of BK receptor 2, B2R (Montana & Sontheimer, 2011), previously correlated to higher grade of tumor (Raidoo et al., 1999). Activation of B2R induces increased motility and invasive properties (Montana & Sontheimer, 2011). Consequently cells migrate toward higher BK concentrations alias blood vessels, as shown in acute slice preparations where BK gradient significantly increased the number of GBM cells attached to blood vessels and boosted glioma infiltration into the tissue (Montana & Sontheimer, 2011). When preparations were treated with FDA approved B2R blocker icatibant (Cicardi et al., 2010) et al. 2010) all the effects of BK were significantly reduced (Montana & Sontheimer, 2011). Bearing in mind that BK may act on normal brain cells such as astrocytes through the same signaling pathway and induce release of various gliotransmitters, as well as increased secretion of MMPs (Parpura et al., 2012), it is appealing to speculate that this novel B2R-mediated GBM-blood vessel signaling pathway may represent a point for adjuvant therapy intervention.
Glioma cells secrete a number of cytokines to attract microglial cells to a tumor mass which is estimated to be around 30%. Microglia harbored within tumor seem to be activated by morphological appearance (Graeber et al., 2002). However, not only their immunological functions are suppressed (Graeber et al., 2002), but the cells support glioma growth and spread by glioma-mediated increase in MT1-MMP expression and activity in microglia (Markovic et al., 2005). The effects were reduced using clinically approved antibiotic minocycline in an experimental mouse model (Markovic et al., 2011), opening new venues in exploring therapeutic treatments.
As GBM infiltrates and grows in the surrounding healthy tissue, it adapts to a new environment and a need for neovascularization becomes apparent. GSC, in addition to its potential to produce heterogeneous cell population and ability to invade, pushes the formation of new vessels to ensure blood supply and sufficient nutrients by producing high levels of VEGF (Bao et al., 2006). Therapy with bevacizumab, a monoclonal antibody that targets VEGF to inhibit angiogenesis and treat recurrent neoplasm was developed (Cohen et al., 2009). While it reduced the tumor size, successful treatment was limited, perhaps because surviving cells develop and/or up-regulate alternative pathways to bypass inhibitory effects of bevacizumab (Lathia et al., 2015).
Concluding remarks
Clearly, interactions between glioma cells and their environment are numerous and very complex. Detached glioma cells are exposed to and respond to a wide range of signaling molecules. Cross-talks of glioma cells with healthy cells, such are endothelial cells and microglia, open new avenues to explore novel therapeutic targets. The lack of experimental models, especially in the context of the microenvironment that would completely recapitulate the disease, is balanced by the discovery of effective therapies; many potential drugs fail in a Clinical Phase. Despite enormous amount of data on GBM generated by –omics approach which broaden our knowledge of GBM biology in the last decade, a breakthrough in therapeutic success did not follow. It became evident that there are not only huge variations between gliomas in different patients, but that each of GBM is very heterogeneous within themselves. That fact underlines the need for a more personalized approach in treating the disease, as well as considering the introduction of novel, adjuvant therapeutics that could affect various interactions of glioma with normal brain cells to complement existing treatments of gliomas. Until then, we will be facing similar obstacles as decades ago in taming the disease, and will be stranded in respect to enhancing the quality of patients’ lives and increasing survival.
| Attachment | Size |
|---|---|
| 427.69 KB |
BAO S., WU Q., SATHORNSUMETEE S., HAO Y., LI Z., HJELMELAND A.B., SHI Q., MCLENDON R.E., BIGNER D.D. & RICH J.N. (2006). Stem cell-like glioma cells promote tumor angiogenesis through vascular endothelial growth factor. Cancer Res 66, 7843-7848.
BEADLE C., ASSANAH M.C., MONZO P., VALLEE R., ROSENFELD S.S. & CANOLL P. (2008). The role of myosin II in glioma invasion of the brain. Mol Biol Cell 19, 3357-3368.
BRENNAN C.W., VERHAAK R.G., MCKENNA A., CAMPOS B., NOUSHMEHR H., SALAMA S.R., ZHENG S., CHAKRAVARTY D., SANBORN J.Z., BERMAN S.H., BEROUKHIM R., BERNARD B., WU C.J., GENOVESE G., SHMULEVICH I., BARNHOLTZ-SLOAN J., ZOU L., VEGESNA R., SHUKLA S.A., CIRIELLO G., YUNG W.K., ZHANG W., SOUGNEZ C., MIKKELSEN T., ALDAPE K., BIGNER D.D., VAN MEIR E.G., PRADOS M., SLOAN A., BLACK K.L., ESCHBACHER J., FINOCCHIARO G., FRIEDMAN W., ANDREWS D.W., GUHA A., IACOCCA M., O’NEILL B.P., FOLTZ G., MYERS J., WEISENBERGER D.J., PENNY R., KUCHERLAPATI R., PEROU C.M., HAYES D.N., GIBBS R., MARRA M., MILLS G.B., LANDER E., SPELLMAN P., WILSON R., SANDER C., WEINSTEIN J., MEYERSON M., GABRIEL S., LAIRD P.W., HAUSSLER D., GETZ G., CHIN L. & NETWORK T.R. (2013). The somatic genomic landscape of glioblastoma. Cell 155, 462-477.
BUCCIONE R., CALDIERI G. & AYALA I. (2009). Invadopodia: specialized tumor cell structures for the focal degradation of the extracellular matrix. Cancer Metastasis Rev 28, 137-149.
Cancer Genome Atlas Research N. (2008). Comprehensive genomic characterization defines human glioblastoma genes and core pathways. Nature 455, 1061-1068.
CHAN X.H., NAMA S., GOPAL F., RIZK P., RAMASAMY S., SUNDARAM G., OW G.S., IVSHINA A.V., TANAVDE V., HAYBAECK J., KUZNETSOV V. & SAMPATH P. (2012). Targeting glioma stem cells by functional inhibition of a prosurvival oncomiR-138 in malignant gliomas. Cell Rep 2, 591-602.
CHEN J., LI Y., YU T.S., MCKAY R.M., BURNS D.K., KERNIE S.G. & PARADA L.F. (2012). A restricted cell population propagates glioblastoma growth after chemotherapy. Nature 488, 522-526.
CHUDNOVSKY Y., KIM D., ZHENG S., WHYTE W.A., BANSAL M., BRAY M.A., GOPAL S., THEISEN M.A., BILODEAU S., THIRU P., MUFFAT J., YILMAZ O.H., MITALIPOVA M., WOOLARD K., LEE J., NISHIMURA R., SAKATA N., FINE H.A., CARPENTER A.E., SILVER S.J., VERHAAK R.G., CALIFANO A., YOUNG R.A., LIGON K.L., MELLINGHOFF I.K., ROOT D.E., SABATINI D.M., HAHN W.C. & CHHEDA M.G. (2014). ZFHX4 interacts with the NuRD core member CHD4 and regulates the glioblastoma tumor-initiating cell state. Cell Rep 6, 313-324.
CICARDI M., BANERJI A., BRACHO F., MALBRAN A., ROSENKRANZ B., RIEDL M., BORK K., LUMRY W., ABERER W., BIER H., BAS M., GREVE J., HOFFMANN T.K., FARKAS H., RESHEF A., RITCHIE B., YANG W., GRABBE J., KIVITY S., KREUZ W., LEVY R.J., LUGER T., OBTULOWICZ K., SCHMID-GRENDELMEIER P., BULL C., SITKAUSKIENE B., SMITH W.B., TOUBI E., WERNER S., ANNE S., BJORKANDER J., BOUILLET L., CILLARI E., HUREWITZ D., JACOBSON K.W., KATELARIS C.H., MAURER M., MERK H., BERNSTEIN J.A., FEIGHERY C., FLOCCARD B., GLEICH G., HEBERT J., KAATZ M., KEITH P., KIRKPATRICK C.H., LANGTON D., MARTIN L., PICHLER C., RESNICK D., WOMBOLT D., FERNANDEZ ROMERO D.S., ZANICHELLI A., ARCOLEO F., KNOLLE J., KRAVEC I., DONG L., ZIMMERMANN J., ROSEN K. & FAN W.T. (2010). Icatibant, a new bradykinin-receptor antagonist, in hereditary angioedema. N Engl J Med 363, 532-541.
CLEMENT V., SANCHEZ P., DE TRIBOLET N., RADOVANOVIC I. & RUIZ I. ALTABA A. (2007). HEDGEHOG-GLI1 signaling regulates human glioma growth, cancer stem cell self-renewal, and tumorigenicity. Curr Biol 17, 165-172.
COHEN M.H., SHEN Y.L., KEEGAN P. & PAZDUR R. (2009). FDA drug approval summary: bevacizumab (Avastin) as treatment of recurrent glioblastoma multiforme. Oncologist 14, 1131-1138.
CUDDAPAH V.A., ROBEL S., WATKINS S. & SONTHEIMER H. (2014). A neurocentric perspective on glioma invasion. Nat Rev Neurosci 15, 455-465.
FANG X., HUANG Z., ZHOU W., WU Q., SLOAN A.E., OUYANG G., MCLENDON R.E., YU J.S., RICH J.N. & BAO S. (2014). The zinc finger transcription factor ZFX is required for maintaining the tumorigenic potential of glioblastoma stem cells. Stem Cells 32, 2033-2047.
FRIEDMANN-MORVINSKI D., BUSHONG E.A., KE E., SODA Y., MARUMOTO T., SINGER O., ELLISMAN M.H. & VERMA I.M. (2012). Dedifferentiation of neurons and astrocytes by oncogenes can induce gliomas in mice. Science 338, 1080-1084.
GALLI R., BINDA E., ORFANELLI U., CIPELLETTI B., GRITTI A., DE VITIS S., FIOCCO R., FORONI C., DIMECO F. & VESCOVI A. (2004). Isolation and characterization of tumorigenic, stem-like neural precursors from human glioblastoma. Cancer Res 64, 7011-7021.
GANGEMI R.M., GRIFFERO F., MARUBBI D., PERERA M., CAPRA M.C., MALATESTA P., RAVETTI G.L., ZONA G.L., DAGA A. & CORTE G. (2009). SOX2 silencing in glioblastoma tumor-initiating cells causes stop of proliferation and loss of tumorigenicity. Stem Cells 27, 40-48.
GRAEBER M.B., SCHEITHAUER B.W. & KREUTZBERG G.W. (2002). Microglia in brain tumors. Glia 40, 252-259.
HEMMATI H.D., NAKANO I., LAZAREFF J.A., MASTERMAN-SMITH M., GESCHWIND D.H., BRONNER-FRASER M. & KORNBLUM H.I. (2003). Cancerous stem cells can arise from pediatric brain tumors. Proc Natl Acad Sci U S A 100, 15178-15183.
HOELZINGER D.B., DEMUTH T. & BERENS M.E. (2007). Autocrine factors that sustain glioma invasion and paracrine biology in the brain microenvironment. J Natl Cancer Inst 99, 1583-1593.
IGNATOVA T.N., KUKEKOV V.G., LAYWELL E.D., SUSLOV O.N., VRIONIS F.D. & STEINDLER D.A. (2002). Human cortical glial tumors contain neural stem-like cells expressing astroglial and neuronal markers in vitro. Glia 39, 193-206.
JIMSHELEISHVILI S., ALSHAREEF A.T., PAPADIMITRIOU K., BREGY A., SHAH A.H., GRAHAM R.M., FERRARO N. & KOMOTAR R.J. (2014). Extracranial glioblastoma in transplant recipients. J Cancer Res Clin Oncol 140, 801-807.
JOSHI K., BANASAVADI-SIDDEGOWDA Y., MO X., KIM S.H., MAO P., KIG C., NARDINI D., SOBOL R.W., CHOW L.M., KORNBLUM H.I., WACLAW R., BEULLENS M. & NAKANO I. (2013). MELK-dependent FOXM1 phosphorylation is essential for proliferation of glioma stem cells. Stem Cells 31, 1051-1063.
KWIATKOWSKA A. & SYMONS M. (2013). Signaling determinants of glioma cell invasion. Adv Exp Med Biol 986, 121-141.
LATHIA J.D., MACK S.C., MULKEARNS-HUBERT E.E., VALENTIM C.L. & RICH J.N. (2015). Cancer stem cells in glioblastoma. Genes Dev 29, 1203-1217.
LIU C., SAGE J.C., MILLER M.R., VERHAAK R.G., HIPPENMEYER S., VOGEL H., FOREMAN O., BRONSON R.T., NISHIYAMA A., LUO L. & ZONG H. (2011). Mosaic analysis with double markers reveals tumor cell of origin in glioma. Cell 146, 209-221.
LOUIS D.N., OHGAKI H., WIESTLER O.D., CAVENEE W.K., BURGER P.C., JOUVET A., SCHEITHAUER B.W. & KLEIHUES P. (2007). The 2007 WHO classification of tumours of the central nervous system. Acta Neuropathol 114, 97-109.
LUI V.C., LUNG S.S., PU J.K., HUNG K.N. & LEUNG G.K. (2010). Invasion of human glioma cells is regulated by multiple chloride channels including ClC-3. Anticancer Res 30, 4515-4524.
LYONS S.A., CHUNG W.J., WEAVER A.K., OGUNRINU T. & SONTHEIMER H. (2007). Autocrine glutamate signaling promotes glioma cell invasion. Cancer Res 67, 9463-9471.
MARCUS H.J., CARPENTER K.L., PRICE S.J. & HUTCHINSON P.J. (2010). In vivo assessment of high-grade glioma biochemistry using microdialysis: a study of energy-related molecules, growth factors and cytokines. J Neurooncol 97, 11-23.
MARKOVIC D.S., GLASS R., SYNOWITZ M., ROOIJEN N. & KETTENMANN H. (2005). Microglia stimulate the invasiveness of glioma cells by increasing the activity of metalloprotease-2. J Neuropathol Exp Neurol 64, 754-762.
MARKOVIC D.S., VINNAKOTA K., VAN ROOIJEN N., KIWIT J., SYNOWITZ M., GLASS R. & KETTENMANN H. (2011). Minocycline reduces glioma expansion and invasion by attenuating microglial MT1-MMP expression. Brain Behav Immun 25, 624-628.
MONTANA V. & SONTHEIMER H. (2011). Bradykinin promotes the chemotactic invasion of primary brain tumors. J Neurosci 31, 4858-4867.
NOUSHMEHR H., WEISENBERGER D.J., DIEFES K., PHILLIPS H.S., PUJARA K., BERMAN B.P., PAN F., PELLOSKI C.E., SULMAN E.P., BHAT K.P., VERHAAK R.G., HOADLEY K.A., HAYES D.N., PEROU C.M., SCHMIDT H.K., DING L., WILSON R.K., VAN DEN BERG D., SHEN H., BENGTSSON H., NEUVIAL P., COPE L.M., BUCKLEY J., HERMAN J.G., BAYLIN S.B., LAIRD P.W., Aldape K & Cancer Genome Atlas Research N. (2010). Identification of a CpG island methylator phenotype that defines a distinct subgroup of glioma. Cancer Cell 17, 510-522.
PARPURA V., HENEKA M.T., MONTANA V., OLIET S.H., SCHOUSBOE A., HAYDON P.G., STOUT R.F., JR., SPRAY D.C., REICHENBACH A., PANNICKE T., PEKNY M., PEKNA M., ZOREC R. & VERKHRATSKY A. (2012). Glial cells in (patho)physiology. J Neurochem 121, 4-27.
PHILLIPS H.S., KHARBANDA S., CHEN R., FORREST W.F., SORIANO R.H., WU T.D., MISRA A., NIGRO J.M., COLMAN H., SOROCEANU L., WILLIAMS P.M., MODRUSAN Z., FEUERSTEIN B.G. & ALDAPE K. (2006). Molecular subclasses of high-grade glioma predict prognosis, delineate a pattern of disease progression, and resemble stages in neurogenesis. Cancer Cell 9, 157-173.
RAIDOO D.M., SAWANT S., MAHABEER R. & BHOOLA K.D. (1999). Kinin receptors are expressed in human astrocytic tumour cells. Immunopharmacology 43, 255-263.
RHEINBAY E., SUVA M.L., GILLESPIE S.M., WAKIMOTO H., PATEL A.P., SHAHID M., OKSUZ O., RABKIN S.D., MARTUZA R.L., RIVERA M.N., LOUIS D.N., KASIF S., CHI A.S. & BERNSTEIN B.E. (2013). An aberrant transcription factor network essential for Wnt signaling and stem cell maintenance in glioblastoma. Cell Rep 3, 1567-1579.
SANAI N., ALVAREZ-BUYLLA A. & BERGER M.S. (2005). Neural stem cells and the origin of gliomas. N Engl J Med 353, 811-822.
SHERRY M.M., REEVES A., WU J.K. & COCHRAN B.H. (2009). STAT3 is required for proliferation and maintenance of multipotency in glioblastoma stem cells. Stem Cells 27, 2383-2392.
SINGH S.K., HAWKINS C., CLARKE I.D., SQUIRE J.A., BAYANI J., HIDE T., HENKELMAN R.M., CUSIMANO M.D. & DIRKS P.B. (2004). Identification of human brain tumour initiating cells. Nature 432, 396-401.
SOROCEANU L., GILLESPIE Y., KHAZAELI M.B. & SONTHEIMER H. (1998). Use of chlorotoxin for targeting of primary brain tumors. Cancer Res 58, 4871-4879.
SOROCEANU L., MANNING T.J., JR. & SONTHEIMER H. (1999). Modulation of glioma cell migration and invasion using Cl- and K+ ion channel blockers. J Neurosci 19, 5942-5954.
SUGIARTO S., PERSSON A.I., MUNOZ E.G., WALDHUBER M., LAMAGNA C., ANDOR N., HANECKER P., AYERS-RINGLER J., PHILLIPS J., SIU J., LIM D.A., VANDENBERG S., STALLCUP W., BERGER M.S., BERGERS G., WEISS W.A. & PETRITSCH C. (2011). Asymmetry-defective oligodendrocyte progenitors are glioma precursors. Cancer Cell 20, 328-340.
UCHIDA N., BUCK D.W., HE D., REITSMA M.J., MASEK M., PHAN T.V., TSUKAMOTO A.S., GAGE F.H. & WEISSMAN I.L. (2000). Direct isolation of human central nervous system stem cells. Proc Natl Acad Sci U S A 97, 14720-14725.
VEHLOW A. & CORDES N. (2013). Invasion as target for therapy of glioblastoma multiforme. Biochim Biophys Acta 1836, 236-244.
VERGINELLI F., PERIN A., DALI R., FUNG K.H., LO R., LONGATTI P., GUIOT M.C., DEL MAESTRO R.F., ROSSI S., DI PORZIO U., STECHISHIN O., WEISS S. & STIFANI S. (2013). Transcription factors FOXG1 and Groucho/TLE promote glioblastoma growth. Nat Commun 4, 2956.
WANG J., WANG H., LI Z., WU Q., LATHIA J.D., MCLENDON R.E., HJELMELAND A.B. & RICH J.N. (2008). c-Myc is required for maintenance of glioma cancer stem cells. PLoS One 3, e3769.
WISE D.R. & THOMPSON C.B. (2010). Glutamine addiction: a new therapeutic target in cancer. Trends Biochem Sci 35, 427-433.
WURDAK H., ZHU S., ROMERO A., LORGER M., WATSON J., CHIANG C.Y., ZHANG J., NATU V.S., LAIRSON L.L., WALKER J.R., TRUSSELL C.M., HARSH G.R., VOGEL H., FELDING-HABERMANN B., ORTH A.P., MIRAGLIA L.J., RINES D.R., SKIRBOLL S.L. & SCHULTZ P.G. (2010). An RNAi screen identifies TRRAP as a regulator of brain tumor-initiating cell differentiation. Cell Stem Cell 6, 37-47.
XIE Q., MITTAL S. & BERENS M.E. (2014). Targeting adaptive glioblastoma: an overview of proliferation and invasion. Neuro Oncol 16, 1575-1584.
ZAGZAG D., ESENCAY M., MENDEZ O., YEE H., SMIRNOVA I., HUANG Y., CHIRIBOGA L., LUKYANOV E., LIU M. & NEWCOMB E.W. (2008). Hypoxia- and vascular endothelial growth factor-induced stromal cell-derived factor-1alpha/CXCR4 expression in glioblastomas: one plausible explanation of Scherer’s structures. Am J Pathol 173, 545-560.
ZBINDEN M., DUQUET A., LORENTE-TRIGOS A., NGWABYT S.N., BORGES I. & RUIZ I. ALTABA A. (2010). NANOG regulates glioma stem cells and is essential in vivo acting in a cross-functional network with GLI1 and p53. EMBO J 29, 2659-2674.
ZHENG H., YING H., YAN H., KIMMELMAN A.C., HILLER D.J., CHEN A.J., PERRY S.R., TONON G., CHU G.C., DING Z., STOMMEL J.M., DUNN K.L., WIEDEMEYER R., YOU M.J., BRENNAN C., WANG Y.A., LIGON K.L., WONG W.H., CHIN L. & DEPINHO R.A. (2008). p53 and Pten control neural and glioma stem/progenitor cell renewal and differentiation. Nature 455, 1129-1133.
ZIMMERMANN D.R. & DOURS-ZIMMERMANN M.T. (2008). Extracellular matrix of the central nervous system: from neglect to challenge. Histochem Cell Biol 130, 635-653.