Classically, the central nervous system (CNS) was considered to contain neurons and three main types of glial cells - astrocytes, oligodendrocytes, and microglia. Now, it has been clearly established that NG2-glia are a fourth glial cell type that are defined by their expression of the NG2 chondroitin sulfate proteoglycan (Cspg4). NG2-glia are also known as oligodendrocyte precursor cells (OPCs) and express the alpha receptor for platelet-derived growth factor (Pdgfra) as well as other oligodendrocyte lineage markers. NG2-glia are most numerous during CNS development when they are responsible for massive generation of oligodendrocytes, the myelin-forming cells of the CNS. A significant population of NG2-glia persist in the adult CNS, where they generate oligodendrocytes throughout life. A unique feature of NG2-glia is that they receive synaptic inputs from neurons and are able to respond rapidly to neurotransmission via their specific ion channel and receptor profiles. Moreover, synaptic and neuronal integrity depend on NG2-glia. Notably, concomitant disruption of NG2-glia, myelin and neurotransmission are key features of many neuropathologies, including Multiple Sclerosis and Alzheimer’s disease (AD). The fact that neurotransmission both regulates and is reliant on NG2-glia and myelin raises the ‘chicken and egg’ question of what comes first – disruption of NG2-glia/myelin or synapses/neurons. It is more useful to think of neurons, NG2-glia and oligodendrocytes/myelin as being functionally integrated and interdependent units, whereby disruption of any one can result in a vicious cycle with potentially devastating effects on CNS function.
Introduction
NG2-glia are defined by their expression of the chondroitin sulphate proteoglycan NG2 (cspg4) and are identified using antibodies against NG2 (Fig. 1) (Stallcup, 1981; Levine & Card, 1987; Stallcup & Beasley, 1987). Most researchers consider NG2-glia to be oligodendrocyte progenitor cells (OPCs), since they express PDGFαR and other early oligodendrocyte lineage transcription factors, such as Sox10 and Olig2, and NG2+/PDGFαR+ cells generate oligodendrocytes in the developing and adult CNS (Rivers et al., 2008; Kang et al., 2010; Zhu et al., 2011). Studies in NG2 null mice show the NG2 proteoglycan promotes OPC proliferation and myelination, most likely by enhancement of growth factor signalling or mediation of OPC interaction with unmyelinated axons (Kucharova & Stallcup, 2010). NG2-glia are a ubiquitous population throughout adult white and grey matter (Fig. 1A), with heterogeneous phenotypic and physiological properties and many do not appear to generate oligodendrocytes (Dimou et al., 2008; Karram et al., 2008; Huang et al., 2014). In addition to oligodendrocyte regeneration, studies in null mice show NG2-glia are important for counteracting neurological deficits and adverse glial responses following injury (Huang et al., 2015). A unique feature of NG2-glia is that they form synapses with neurons and display spontaneous and evoked synaptic currents (Bergles et al., 2010; Dimou & Gallo, 2015), although expression of the NG2 proteoglycan per se is not required for formation of neuron-NG2-glial cell synapses (Passlick et al., 2016). The functions of NG2-glia at synapses is an evolving story, but it is becoming increasingly clear that synaptic activity acting via a range of neurotransmitter receptors and ion channels regulates the proliferation and differentiation of NG2-glia (Butt et al., 2014; Dimou & Gallo, 2015). Notably, there is bidirectional cross-talk between NG2-glia and the neuronal network, whereby NG2-glia regulate information processing at neuronal synapses and their ablation causes deficits in glutamatergic neurotransmission and depressive-like behaviour in mice (Sakry et al., 2014; Birey et al., 2015). Furthermore, the continued generation of oligodendrocytes by NG2-glia throughout life has important implications for rapid communication and cognitive function (Young et al., 2013; McKenzie et al., 2014), but the myelin sheath is not just an inert insulating structure and is essential for the long-term integrity of myelinated axons (Hirrlinger & Nave, 2014). Notably, glutamatergic neurotransmission, NG2-glia, myelin loss and axon degeneration are inextricably involved in multiple pathologies, including traumatic injury, Multiple Sclerosis (MS), amyotrophic lateral sclerosis sclerosis (ALS), Alzheimer’s disease (AD), stroke and leukodystrophies (Butt et al., 2014; Fern et al., 2014; Rivera et al., 2015). Hence, understanding the physiological properties of NG2-glia is important to developing new cellular and molecular targets aimed at maintaining neuronal and myelin integrity.
Morphology and distribution of ng2-glia
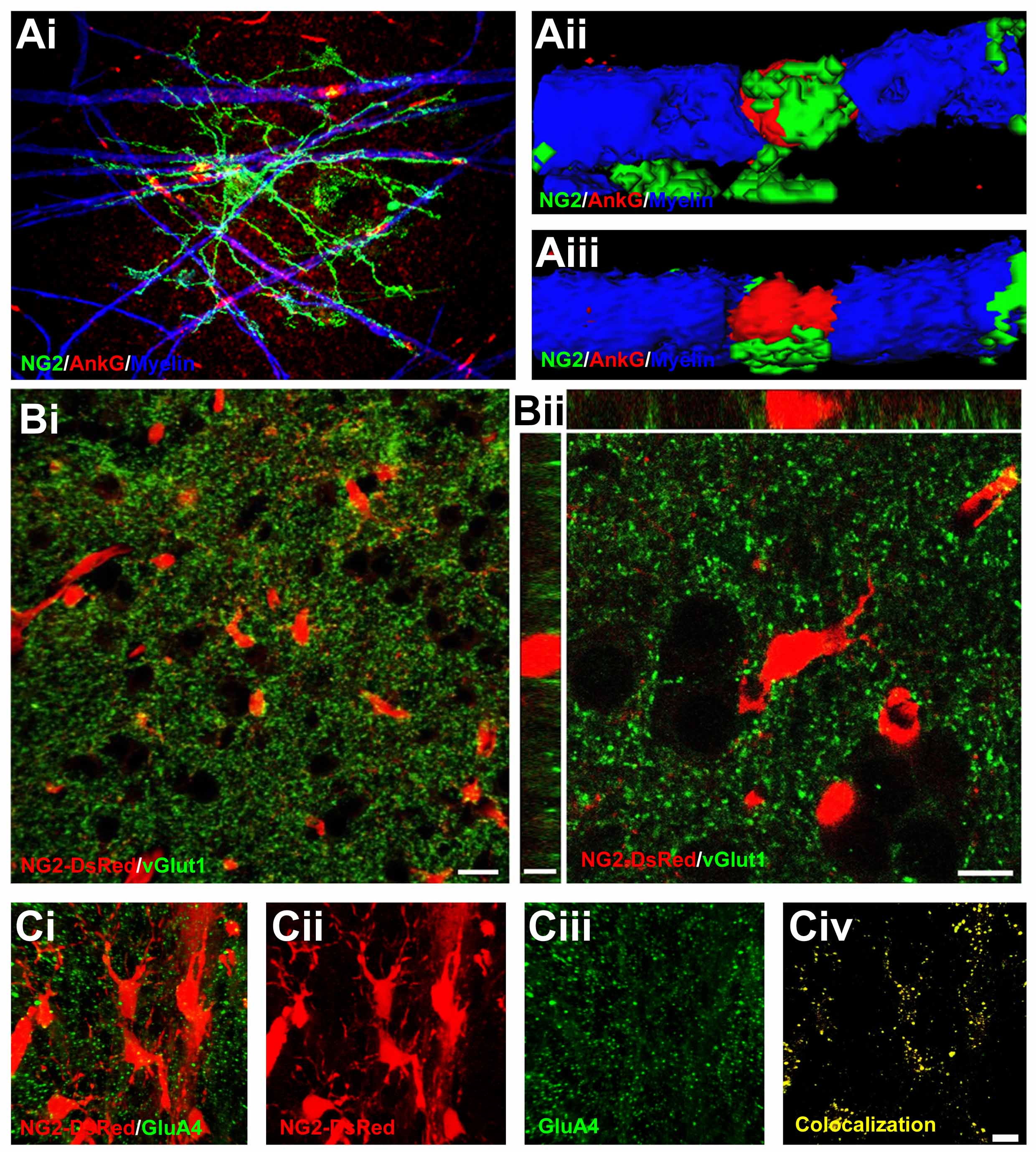
NG2-glia constitute 8-9% of total cells in the white matter and 2-3% of total cells in the grey matter (Hill & Nishiyama, 2014). NG2-glia have a multi-processed morphology, with their process domains or territories overlapping slightly at their edges (Fig. 1A)(Butt et al., 1999; Leoni et al., 2009; Wigley & Butt, 2009; Hamilton et al., 2010; Xu et al., 2014). EM studies show that the processes of NG2-glia contact synapses in grey matter and electrophysiological studies show they display both GABAergic and glutamatergic inputs (Bergles et al., 2010). During development, there is a postnatal switch from synaptic to extrasynaptic transmission in NG2-glia and in the adult volume transmission is likely to be most important (Velez-Fort et al., 2010). NG2-glia have overlapping domains with astrocytes and there is extensive interdigitation of their processes (Fig. 1B); indeed NG2-glia and astrocytes appear to serve the same neurons and are closely apposed at the same synapses (Fig. 1C) (Leoni et al., 2009; Wigley & Butt, 2009; Hamilton et al., 2010; Xu et al., 2014). However, electrophysiological studies indicate NG2-glia and astrocytes are not coupled through gap junctions and neither electrical nor tracer coupling could be detected from NG2 glia-NG2 glia or astrocyte-NG2 glia pairs (Xu et al., 2014). In myelinated fibre tracts, NG2-glial cell processes contact nodes of Ranvier and respond to axonally released glutamate and ATP released from astrocytes (Fig. 2A) (Butt et al., 1999; Kukley et al., 2007; Ziskin et al., 2007; Hamilton et al., 2010).
Figure 1. Neuron-glial interrelations of NG2-glia in the adult brain. Confocal micrographs of sections of mouse brain immunolabelled with anti-NG2 antibody, (A) with myelin basic protein (MBP) to identify myelinated axons, (B, C) enhanced green fluorescent protein (eGFP) driven by glial fibrillary acidic protein (GFAP) to identify astrocytes, and (C) synaptophysin to identify neuronal synapses. NG2-glia (green in A, red in B,C) are abundant throughout grey matter and form circular domains within which they contact multiple neurons (blue, A), myelinated axons (red, A), blood vessels, astrocytes (green, B, C) and synapses (blue, C). The images in (C) are from the hippocampus and illustrate an astrocyte (green) and NG2-glial cell (red) with directly apposed cell bodies and intertwining processes that are associated with innumerable synapses labelled for the presynaptic protein synaptophysin (blue); (Civ) is a single z-section and (Cv) is an isosurface rendered view illustrating the immediate relationships between NG2-glia and astrocytes with multiple synapses.
Neurotransmitter receptors in ng2-glia
NG2-glia express a wide range of neurotransmitter receptors and ion channels (Larson et al., 2015). However, a ubiquitous feature of most NG2-glia studied is their close apposition to glutamatergic synapses (Fig. 2B) and their prominent expression of AMPA-type glutamate receptors (Fig. 2C). NG2-glia co-express calcium-impermeable AMPA-R containing GluA2 and calcium-permeable AMPA-R containing GluA4 (Seifert et al., 1997; Bergles et al., 2000; Ge et al., 2006; Hamilton et al., 2010; Larson et al., 2015). NG2-glia also express NMDA receptors, the most highly-expressed subunits being GluN3A and GluN1, with GluN2C and GluN2D being expressed at lower levels (Larson et al., 2015), which endows the NMDA-R with reduced sensitivity to block by Mg2+, suggesting they could contribute to significant Ca2+ influx at resting membrane potential (Karadottir et al., 2005; Ziskin et al., 2007; De Biase et al., 2010). In addition, NG2-glia form synapses with GABAergic neurons and express functional GABAAR (Lin & Bergles, 2004; Velez-Fort et al., 2010; Orduz et al., 2015). Physiological, RT-PCR and RNA-seq analyses agree that NG2-glia express all α subunits with the exception of α6, and functional GABAAR most likely are made up of predominantly of α3, β2/β3 and γ1 subunits (Passlick et al., 2013; Balia et al., 2015; Larson et al., 2015). However, the molecular composition of GABAAR in NG2-glia may be heterogeneous and changes with age. Notably, expression of the γ2 subunit, which is important for synaptic localization, is markedly down-regulated with age and this may underlie the developmental loss of direct GABAergic synaptic contacts in NG2-glia (Velez-Fort et al., 2010; Passlick et al., 2013; Balia et al., 2015). A consistent feature of NG2-glia is that they also express functional purine receptors, with a predominance of P2X7 and P2Y1 subtypes (Agresti et al., 2005a; Agresti et al., 2005b; Hamilton et al., 2010; Butt et al., 2014; Larson et al., 2015).
Ion channels in ng2-glia
NG2-glia have a highly negative resting membrane potential of approximately –90 mV and prominent inward rectifier K+ channels that are open at rest. NG2-glia express Kir4.1 channels and they are the dominant resting conductance in NG2-glia and determine their strongly negative resting potential (Djukic et al., 2007; Maldonado et al., 2013). Kir4.1 is an inward-rectifying channel expressed by astrocytes and oligodendrocytes (Butt & Kalsi, 2006), and NG2-glia and RNA-Seq transcriptome analysis shows that Kir4.1 (Kcnj10) mRNA is the most highly expressed K+ channel subunit in NG2-glia (Larson et al., 2015). Notably, Kir5.1 specifically forms heteromeric channels with Kir4.1 and is more highly expressed in NG2-glia than in astrocytes, perhaps indicating a relatively greater role for heteromeric channels in NG2-glia. Electrophysiological studies indicate NG2-glia also express A-type and delayed-rectifier K+ channels (Chittajallu et al., 2004), which RNA-seq analysis indicates are likely to comprise mainly Kv4.2/Kv4.3 and Kv 1.6 subtypes (Larson et al., 2015). In addition, NG2-glia express voltage-gated sodium channels, which are likely to be heterogeneous and comprise predominantly of Nav1.3, Nav1.2, Nav1.8, and Nav1.1 (Larson et al., 2015). Upon depolarization, NG2-glia can display one or more Na+ spikes (Chittajallu et al., 2004; Karadottir et al., 2008), but this disappears as the Kv/Nav ratio increases in adult NG2-glia (De Biase et al., 2010; Maldonado et al., 2013). Whole-cell recordings from NG2-glia have revealed the presence of L-type and T-type Ca2+ channels (Haberlandt et al., 2011), which is substantiated by RT-PCR and RNA-seq analyses (Haberlandt et al., 2011; Larson et al., 2015), and Cav1.2 appears to be the principal pore-forming subunit (Cheli et al., 2015).
Figure 2. NG2-glial relations with nodes of Ranvier and glutamatergic synapses. Confocal microscopic images of NG2-glia. (A) Anterior medullary velum of the adult rat immunolabelled for NG2 (green), MBP (blue) and the nodal axolemmal protein AnkG (red), illustrating NG2-glial cell extending processes to contact multiple nodes of Ranvier, the sites of action potential propagation and glutamate release; (Aii, Aiii) are isosurface rendered views illustrating the immediate apposition of NG2-glial cell processes with the node of Ranvier and myelin. (B) Immunolabelling for the presynaptic vesicular glutamate transporter vGLUT1 (green) in the cortex of NG2-DsRed reporter mice in which NG2 drives the expression of DsRed; (Bi) illustrates the high density of synapses within the domains of NG2-glia; (Bii) single z-section (main panel) and x-x (top panel) and y-y (left-hand panel) cross-sectional images illustrating the immediate apposition of glutamatergic synapses on NG2-glial cell somata and process. (C) Immunolabelling for the postsynaptic AMPA-type receptor subunit GluA4 (green) in the cerebellum of NG2-dsRed mice; (Civ) the colocalisation channel indicates GluA4 decorate the somata and processes of NG2-glia.
Neuron-ng2-glial cell signalling
Excitatory postsynaptic currents mediated by AMPA-R are evident in NG2-glia from all brain regions where it has been studied, suggesting that this mode of communication is a conserved property of these glial cells (Bergles et al., 2010). Activation of AMPA-R in NG2-glia stimulates their migration (Gudz et al., 2006), and there is clear evidence that glutamate signalling provides a mechanism of adaptive myelination of electrically active axons (Wake et al., 2011; Gibson et al., 2014). Blockade of AMPA-R has been shown to increase proliferation and differentiation of NG2-glia in cerebellar slice cultures, but did not promote myelination (Fannon et al., 2015), and glutamate neurotransmission is also important in the early stages of remyelination following demyelination (Etxeberria et al., 2010). Similarly, NG2-glia exhibit spontaneous, miniature and evoked GABAAR-mediated currents in the hippocampus, cortex and cerebellum (Maldonado et al., 2011). Blockade of GABAAR promotes proliferation of NG2-glia and inhibits their differentiation, whereas increasing GABAergic transmission has the opposite effect (Zonouzi et al., 2015). Hence, there is growing evidence to support the notion that neurotransmission regulates the proliferation of NG2-glia and their differentiation into myelinating oligodendrocytes.
Recent studies provide evidence that neuron-NG2-glial cell communication is bidirectional and that NG2-glia modulate neuronal function. Ablation of NG2 results in a striking reduction of NMDA receptor-dependent long-term potentiation in pyramidal neurons of the somatosensory cortex and alters the subunit composition of their AMPA receptors (Sakry et al., 2014). Furthermore, NG2-glia express the neuromodulatory protein neuronal Pentraxin 2 (Nptx2/Narp), which on release binds to the GluA2 subunit of neuronal AMPA-R and modulates neuronal signalling (Sakry et al., 2015). Consistent with this, NG2-knockout mice exhibit altered sensorimotor function (Sakry et al., 2014) and ablation of NG2-glia in the prefrontal cortex of the adult brain causes deficits in excitatory glutamatergic neurotransmission and induces depressive-like behaviour in mice (Birey et al., 2015). Furthermore, studies in NG2 knockout mice support a role for NG2-glia in counteracting myelin loss and neurological deficits following demyelination and injury (Kucharova et al., 2011; Huang et al., 2015). Thus, neuron-NG2-glial cell interactions are critical for neuronal integrity and repair.
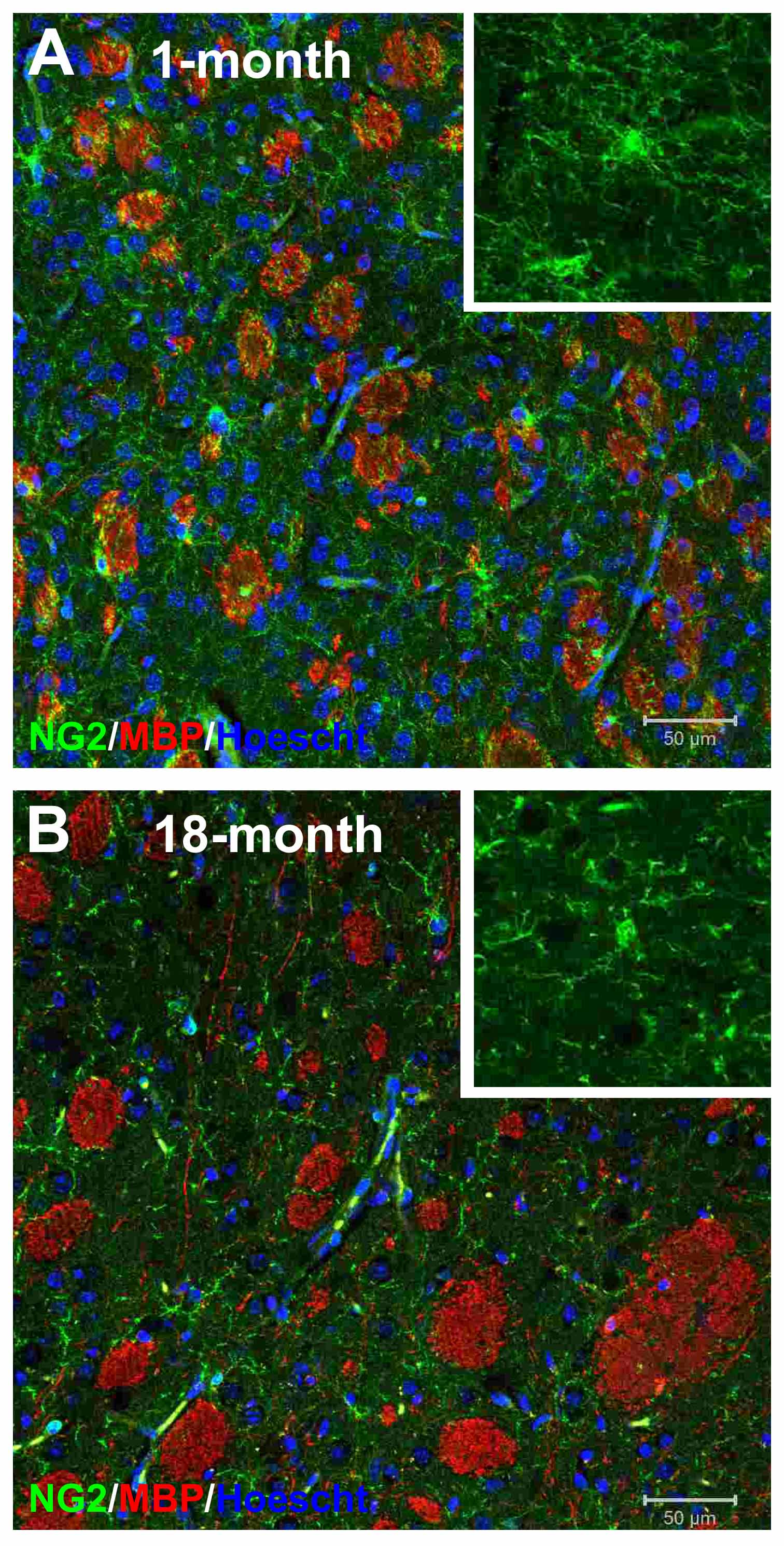
Figure 3. NG2-glia are altered in the ageing mouse brain. Confocal micrographs of striatum immunolabelled for NG2 (green), MBP (red) and counterstained with the nuclear dye Hoescht (blue) in the 1- and 18-month old mouse. There is an apparent decrease in NG2-glia in the ageing brain and higher magnification (insets) indicates a retraction of NG2-glial cell processes in the ageing brain, consistent with a decrease in synaptic connectivity.
Neurotransmission and ng2-glial cell pathology
Excessive activation of glutamate ionotropic receptors results in excitotoxicity and is highly relevant to NG2-glia and oligodendrocytes in multiple pathologies (Back & Rosenberg, 2014; Fern et al., 2014; Haroutunian et al., 2014; Kou & VandeVord, 2014; Lassmann, 2014). Excitotoxicity is often associated with hypoxic-ischemic injury, which occurs in periventricular leukomalacia (PVL) in neonates and in stroke in adults, as well as in vascular dementia in the aging brain. NG2-glia and immature oligodendrocytes are very sensitive to hypoxia-ischemia due to their expression of AMPA receptors (Back & Rosenberg, 2014). Notably, synapses between NG2-glia and axons are profoundly damaged in PVL models (Shen et al., 2012). In addition, NMDA receptors mediate damage to oligodendrocytes and the myelin sheath (Salter & Fern, 2005; Micu et al., 2006), and NMDA-R blockade with memantine attenuates oligodendrocyte loss in experimental PVL (Manning et al., 2008). Primary and/or secondary alterations in glutamate signalling also contribute to the pathology of MS, the major demyelinating disease of the CNS (Lassmann, 2014). Genome-wide association screening studies have identified associated alleles in AMPA receptor genes in MS patients who exhibited the highest levels of glutamate and brain volume loss (Baranzini et al., 2010).
Recent papers highlight the importance of glutamate signalling and myelin loss in neuropsychiatric diseases and AD (Haroutunian et al., 2014; Rivera et al., 2015). Myelin loss has been identified a key feature of AD in humans and animal models, resulting in the loss of synchronization of higher cognitive functions. Notably, there are marked changes in NG2-glia in the ageing brain (Fig. 3) and these are accelerated in the 3xTg-AD model (Rivera et al., 2015). Genomic analysis of the aging mouse optic nerve, a typical CNS white matter tract, indicated disruption of NG2-glia and myelin-related genes were correlated with marked changes in glutamatergic neurotransmission (Rivera et al., 2015). Moreover, myelin defects are a prominent feature of human AD and animal models of AD and coincide with impairment of glutamatergic activity (Oddo et al., 2003; Desai et al., 2009). These studies are consistent with changes in synaptic glutamate signalling being interwoven with disruption of NG2-glia and myelin loss in the ageing brain. It is notable that our examination of the ageing brain (Fig. 3) and the 3xTg-AD (Rivera et al., 2015) indicates an apparent atrophy of NG2-glia, consistent with a retraction of synaptic processes. Since glutamatergic signalling regulates the proliferation and differentiation of NG2-glia, the potential loss of synaptic connections would significantly alter their regenerative capacity, contributing to myelin loss and cognitive decline. Furthermore, recent evidence that NG2-glia are important for the maintenance of glutamatergic synapses suggests that loss of NG2-glial cell synaptic connectivity may have a primary importance in impairment of synaptic signalling in AD.
Summary and conclusions
NG2-glia are prominent throughout CNS white and grey matter. One of their key functions is to generate oligodendrocytes throughout life, which is critical for normal brain function and repair following myelin loss that is a feature of most neuropathologies. NG2-glia express multiple receptors and ion channels, but a key feature is glutamate-mediated synaptic signalling via AMPA receptors. Glutamatergic neurotransmission in NG2-glia regulates their proliferation and differentiation, ensuring adequate recruitment of NG2-glia to maintain and replace myelin throughout life. Notably, bidirectional neuron-NG2-glia interactions regulate glutamatergic neurotransmission, information processing and behaviour. Dysregulation of glutamate signalling is implicated in oligodendrocyte pathology and in myelin loss. Furthermore, the myelin sheath is essential for the long-term integrity of myelinated axons and neurons. This leads to the ‘chicken and egg’ question in the title of this review. Which comes first? Is it the neurocentric view of disrupted neurotransmission causing a secondary reaction in glia with consequent damage to axons and neurons? Or, could altered functionality of NG2-glia be a primary event in disruption of neurotransmission and myelin loss? These processes are inextricably linked and disruption of any of the cellular constituents will affect the others, hence targeting NG2-glia may help break the cycle of synaptic disruption and oligodendrocyte/myelin loss.
| Attachment | Size |
|---|---|
| 909.28 KB |
The authors would like to acknowledge funders of their research - the BBSRC, Anatomical Society, University of Portsmouth Research Development Fund and IBBS.
AGRESTI C., MEOMARTINI M.E., AMADIO S., AMBROSINI E., SERAFINI B., FRANCHINI L., VOLONTE C., ALOISI F. & VISENTIN S. (2005a) Metabotropic P2 receptor activation regulates oligodendrocyte progenitor migration and development. Glia, 50, 132-144.
AGRESTI C., MEOMARTINI M.E., AMADIO S., AMBROSINI E., VOLONTE C., ALOISI F. & VISENTIN S. (2005b) ATP regulates oligodendrocyte progenitor migration, proliferation, and differentiation: involvement of metabotropic P2 receptors. Brain Res Brain Res Rev, 48, 157-165.
BACK S.A. & ROSENBERG P.A. (2014) Pathophysiology of glia in perinatal white matter injury. Glia, 62, 1790-1815.
BALIA M., VELEZ-FORT M., PASSLICK S., SCHAFER C., AUDINAT E., STEINHAUSER C., SEIFERT G. & ANGULO M.C. (2015) Postnatal down-regulation of the GABAA receptor gamma2 subunit in neocortical NG2 cells accompanies synaptic-to-extrasynaptic switch in the GABAergic transmission mode. Cereb Cortex, 25, 1114-1123.
BARANZINI S.E., SRINIVASAN R., KHANKHANIAN P., OKUDA D.T., NELSON S.J., MATTHEWS P.M., HAUSER S.L., OKSENBERG J.R. & PELLETIER D. (2010) Genetic variation influences glutamate concentrations in brains of patients with multiple sclerosis. Brain, 133, 2603-2611.
BERGLES D.E., JABS R. & STEINHAUSER C. (2010) Neuron-glia synapses in the brain. Brain Res Rev, 63, 130-137.
BERGLES D.E., ROBERTS J.D., SOMOGYI P. & JAHR C.E. (2000) Glutamatergic synapses on oligodendrocyte precursor cells in the hippocampus. Nature, 405, 187-191.
BIREY F., KLOC M., CHAVALI M., HUSSEIN I., WILSON M., CHRISTOFFEL D.J., CHEN T., FROHMAN M.A., ROBINSON J.K., RUSSO S.J., MAFFEI A. & AGUIRRE A. (2015) Genetic and Stress-Induced Loss of NG2 Glia Triggers Emergence of Depressive-like Behaviors through Reduced Secretion of FGF2. Neuron, 88, 941-956.
BUTT A.M., DUNCAN A., HORNBY M.F., KIRVELL S.L., HUNTER A., LEVINE J.M. & BERRY M. (1999) Cells expressing the NG2 antigen contact nodes of Ranvier in adult CNS white matter. Glia, 26, 84-91.
BUTT A.M., FERN R.F. & MATUTE C. (2014) Neurotransmitter signaling in white matter. Glia, 62, 1762-1779.
BUTT A.M., HAMILTON N., HUBBARD P., PUGH M. & IBRAHIM M. (2005) Synantocytes: the fifth element. J Anat, 207, 695-706.
BUTT A.M. & KALSI A. (2006) Inwardly rectifying potassium channels (Kir) in central nervous system glia: a special role for Kir4.1 in glial functions. J Cell Mol Med, 10, 33-44.
CHELI V.T., SANTIAGO GONZALEZ D.A., SPREUER V. & PAEZ P.M. (2015) Voltage-gated Ca2+ entry promotes oligodendrocyte progenitor cell maturation and myelination in vitro. Exp Neurol, 265, 69-83.
CHITTAJALLU R., AGUIRRE A. & GALLO V. (2004) NG2-positive cells in the mouse white and grey matter display distinct physiological properties. J Physiol, 561, 109-122.
DE BIASE L.M., NISHIYAMA A. & BERGLES D.E. (2010) Excitability and synaptic communication within the oligodendrocyte lineage. J Neurosci, 30, 3600-3611.
DESAI M.K., SUDOL K.L., JANELSINS M.C., MASTRANGELO M.A., FRAZER M.E. & BOWERS W.J. (2009) Triple-transgenic Alzheimer’s disease mice exhibit region-specific abnormalities in brain myelination patterns prior to appearance of amyloid and tau pathology. Glia, 57, 54-65.
DIMOU L. & GALLO V. (2015) NG2-glia and their functions in the central nervous system. Glia, 63, 1429-1451.
DIMOU L., SIMON C., KIRCHHOFF F., TAKEBAYASHI H. & GOTZ M. (2008) Progeny of Olig2-expressing progenitors in the gray and white matter of the adult mouse cerebral cortex. J Neurosci, 28, 10434-10442.
DJUKIC B., CASPER K.B., PHILPOT B.D., CHIN L.S. & MCCARTHY K.D. (2007) Conditional knock-out of Kir4.1 leads to glial membrane depolarization, inhibition of potassium and glutamate uptake, and enhanced short-term synaptic potentiation. J Neurosci, 27, 11354-11365.
ETXEBERRIA A., MANGIN J.M., AGUIRRE A. & GALLO V. (2010) Adult-born SVZ progenitors receive transient synapses during remyelination in corpus callosum. Nat Neurosci, 13, 287-289.
FANNON J., TARMIER W. & FULTON D. (2015) Neuronal activity and AMPA-type glutamate receptor activation regulates the morphological development of oligodendrocyte precursor cells. Glia, 63, 1021-1035.
FERN R.F., MATUTE C. & STYS P.K. (2014) White matter injury: Ischemic and nonischemic. Glia, 62, 1780-1789.
GE W.P., YANG X.J., ZHANG Z., WANG H.K., SHEN W., DENG Q.D. & DUAN S. (2006) Long-term potentiation of neuron-glia synapses mediated by Ca2+ -permeable AMPA receptors. Science, 312, 1533-1537.
GIBSON E.M., PURGER D., MOUNT C.W., GOLDSTEIN A.K., LIN G.L., WOOD L.S., INEMA I., MILLER S.E., BIERI G., ZUCHERO J.B., BARRES B.A., WOO P.J., VOGEL H. & MONJE M. (2014) Neuronal activity promotes oligodendrogenesis and adaptive myelination in the mammalian brain. Science, 344, 1252304.
GUDZ T.I., KOMURO H. & MACKLIN W.B. (2006) Glutamate stimulates oligodendrocyte progenitor migration mediated via an alphav integrin/myelin proteolipid protein complex. J Neurosci, 26, 2458-2466.
HABERLANDT C., DEROUICHE A., WYCZYNSKI A., HASELEU J., POHLE J., KARRAM K., TROTTER J., SEIFERT G., FROTSCHER M., STEINHAUSER C. & JABS R. (2011) Gray matter NG2 cells display multiple Ca2+ -signaling pathways and highly motile processes. PLoS One, 6, e17575.
HAMILTON N., VAYRO S., WIGLEY R. & BUTT A.M. (2010) Axons and astrocytes release ATP and glutamate to evoke calcium signals in NG2-glia. Glia, 58, 66-79.
HAROUTUNIAN V., KATSEL P., ROUSSOS P., DAVIS K.L., ALTSHULER L.L. & BARTZOKIS G. (2014) Myelination, oligodendrocytes, and serious mental illness. Glia, 62, 1856-1877.
HILL R.A. & NISHIYAMA A. (2014) NG2 cells (polydendrocytes): listeners to the neural network with diverse properties. Glia, 62, 1195-1210.
HIRRLINGER J. & NAVE K.A. (2014) Adapting brain metabolism to myelination and long-range signal transduction. Glia, 62, 1749-1761.
HUANG C., SAKRY D., MENZEL L., DANGEL L., SEBASTIANI A., KRAMER T., KARRAM K., ENGELHARD K., TROTTER J. & SCHAFER M.K. (2015) Lack of NG2 exacerbates neurological outcome and modulates glial responses after traumatic brain injury. Glia.
HUANG W., ZHAO N., BAI X., KARRAM K., TROTTER J., GOEBBELS S., SCHELLER A. & KIRCHHOFF F. (2014) Novel NG2-CreERT2 knock-in mice demonstrate heterogeneous differentiation potential of NG2 glia during development. Glia, 62, 896-913.
KANG S.H., FUKAYA M., YANG J.K., ROTHSTEIN J.D. & BERGLES D.E. (2010) NG2+ CNS glial progenitors remain committed to the oligodendrocyte lineage in postnatal life and following neurodegeneration. Neuron, 68, 668-681.
KARADOTTIR R., CAVELIER P., BERGERSEN L.H. & ATTWELL D. (2005) NMDA receptors are expressed in oligodendrocytes and activated in ischaemia. Nature, 438, 1162-1166.
KARADOTTIR R., HAMILTON N.B., BAKIRI Y. & ATTWELL D. (2008) Spiking and nonspiking classes of oligodendrocyte precursor glia in CNS white matter. Nat Neurosci, 11, 450-456.
KARRAM K., GOEBBELS S., SCHWAB M., JENNISSEN K., SEIFERT G., STEINHAUSER C., NAVE K.A. & TROTTER J. (2008) NG2-expressing cells in the nervous system revealed by the NG2-EYFP-knockin mouse. Genesis, 46, 743-757.
KOU Z. & VANDEVORD P.J. (2014) Traumatic white matter injury and glial activation: from basic science to clinics. Glia, 62, 1831-1855.
KUCHAROVA K., CHANG Y., BOOR A., YONG V.W. & STALLCUP W.B. (2011) Reduced inflammation accompanies diminished myelin damage and repair in the NG2 null mouse spinal cord. J Neuroinflammation, 8, 158.
KUCHAROVA K. & STALLCUP W.B. (2010) The NG2 proteoglycan promotes oligodendrocyte progenitor proliferation and developmental myelination. Neuroscience, 166, 185-194.
KUKLEY M., CAPETILLO-ZARATE E. & DIETRICH D. (2007) Vesicular glutamate release from axons in white matter. Nat Neurosci, 10, 311-320.
LARSON V.A., ZHANG Y. & BERGLES D.E. (2015) Electrophysiological properties of NG2 cells: Matching physiological studies with gene expression profiles. Brain Res.
LASSMANN H. (2014) Mechanisms of white matter damage in multiple sclerosis. Glia, 62, 1816-1830.
LEONI G., RATTRAY M. & BUTT A.M. (2009) NG2 cells differentiate into astrocytes in cerebellar slices. Mol Cell Neurosci, 42, 208-218.
LEVINE J.M. & CARD J.P. (1987) Light and electron microscopic localization of a cell surface antigen (NG2) in the rat cerebellum: association with smooth protoplasmic astrocytes. J Neurosci, 7, 2711-2720.
LIN S.C. & BERGLES D.E. (2004) Synaptic signaling between GABAergic interneurons and oligodendrocyte precursor cells in the hippocampus. Nat Neurosci, 7, 24-32.
MALDONADO P.P., VELEZ-FORT M. & ANGULO M.C. (2011) Is neuronal communication with NG2 cells synaptic or extrasynaptic? J Anat, 219, 8-17.
MALDONADO P.P., VELEZ-FORT M., LEVAVASSEUR F. & ANGULO M.C. (2013) Oligodendrocyte precursor cells are accurate sensors of local K+ in mature gray matter. J Neurosci, 33, 2432-2442.
MANNING S.M., TALOS D.M., ZHOU C., SELIP D.B., PARK H.K., PARK C.J., VOLPE J.J. & JENSEN F.E. (2008) NMDA receptor blockade with memantine attenuates white matter injury in a rat model of periventricular leukomalacia. J Neurosci, 28, 6670-6678.
MCKENZIE I.A., OHAYON D., LI H., DE FARIA J.P., EMERY B., TOHYAMA K. & RICHARDSON W.D. (2014) Motor skill learning requires active central myelination. Science, 346, 318-322.
MICU I., JIANG Q., CODERRE E., RIDSDALE A., ZHANG L., WOULFE J., YIN X., TRAPP B.D., MCRORY J.E., REHAK R., ZAMPONI G.W., WANG W. & STYS P.K. (2006) NMDA receptors mediate calcium accumulation in myelin during chemical ischaemia. Nature, 439, 988-992.
ODDO S., CACCAMO A., SHEPHERD J.D., MURPHY M.P., GOLDE T.E., KAYED R., METHERATE R., MATTSON M.P., AKBARI Y. & LAFERLA F.M. (2003) Triple-transgenic model of Alzheimer’s disease with plaques and tangles: intracellular Abeta and synaptic dysfunction. Neuron, 39, 409-421.
ORDUZ D., MALDONADO P.P., BALIA M., VELEZ-FORT M., DE SARS V., YANAGAWA Y., EMILIANI V. & ANGULO M.C. (2015) Interneurons and oligodendrocyte progenitors form a structured synaptic network in the developing neocortex. Elife, 4.
PASSLICK S., GRAUER M., SCHAFER C., JABS R., SEIFERT G. & STEINHAUSER C. (2013) Expression of the gamma2-subunit distinguishes synaptic and extrasynaptic GABA(A) receptors in NG2 cells of the hippocampus. J Neurosci, 33, 12030-12040.
PASSLICK S., TROTTER J., SEIFERT G., STEINHAUSER C. & JABS R. (2016) The NG2 Protein Is Not Required for Glutamatergic Neuron-NG2 Cell Synaptic Signaling. Cereb Cortex, 26, 51-57.
RIVERS L.E., YOUNG K.M., RIZZI M., JAMEN F., PSACHOULIA K., WADE A., KESSARIS N. & RICHARDSON W.D. (2008) PDGFRA/NG2 glia generate myelinating oligodendrocytes and piriform projection neurons in adult mice. Nat Neurosci, 11, 1392-1401.
SAKRY D., NEITZ A., SINGH J., FRISCHKNECHT R., MARONGIU D., BINAME F., PERERA S.S., ENDRES K., LUTZ B., RADYUSHKIN K., TROTTER J. & MITTMANN T. (2014) Oligodendrocyte precursor cells modulate the neuronal network by activity-dependent ectodomain cleavage of glial NG2. PLoS Biol, 12, e1001993.
SAKRY D., YIGIT H., DIMOU L. & TROTTER J. (2015) Oligodendrocyte precursor cells synthesize neuromodulatory factors. PLoS One, 10, e0127222.
SALTER M.G. & FERN R. (2005) NMDA receptors are expressed in developing oligodendrocyte processes and mediate injury. Nature, 438, 1167-1171.
SEIFERT G., REHN L., WEBER M. & STEINHAUSER C. (1997) AMPA receptor subunits expressed by single astrocytes in the juvenile mouse hippocampus. Brain Res Mol Brain Res, 47, 286-294.
SHEN Y., LIU X.B., PLEASURE D.E. & DENG W. (2012) Axon-glia synapses are highly vulnerable to white matter injury in the developing brain. J Neurosci Res, 90, 105-121.
STALLCUP W.B. (1981) The NG2 antigen, a putative lineage marker: immunofluorescent localization in primary cultures of rat brain. Dev Biol, 83, 154-165.
STALLCUP W.B. & BEASLEY L. (1987) Bipotential glial precursor cells of the optic nerve express the NG2 proteoglycan. J Neurosci, 7, 2737-2744.
RIVERA A., VANZULI, I., ARELLANO J.J. & BUTT A. (2016) Decreased regenerative capacity of oligodendrocyte progenitor cells (NG2-glia) in the ageing brain: a vicious cycle of synaptic dysfunction, myelin loss and neuronal disruption? Curr Alzheimer Res.
VELEZ-FORT M., MALDONADO P.P., BUTT A.M., AUDINAT E. & ANGULO M.C. (2010) Postnatal switch from synaptic to extrasynaptic transmission between interneurons and NG2 cells. J Neurosci, 30, 6921-6929.
WAKE H., LEE P.R. & FIELDS R.D. (2011) Control of local protein synthesis and initial events in myelination by action potentials. Science, 333, 1647-1651.
WIGLEY R. & BUTT A.M. (2009) Integration of NG2-glia (synantocytes) into the neuroglial network. Neuron Glia Biol, 5, 21-28.
XU G., WANG W. & ZHOU M. (2014) Spatial organization of NG2 glial cells and astrocytes in rat hippocampal CA1 region. Hippocampus, 24, 383-395.
YOUNG K.M., PSACHOULIA K., TRIPATHI R.B., DUNN S.J., COSSELL L., ATTWELL D., TOHYAMA K. & RICHARDSON W.D. (2013) Oligodendrocyte dynamics in the healthy adult CNS: evidence for myelin remodeling. Neuron, 77, 873-885.
Zhu X., Hill R.A., Dietrich D., Komitova M., Suzuki R. & Nishiyama A. (2011) Age-dependent fate and lineage restriction of single NG2 cells. Development, 138, 745-753.
ZISKIN J.L., NISHIYAMA A., RUBIO M., FUKAYA M. & BERGLES D.E. (2007) Vesicular release of glutamate from unmyelinated axons in white matter. Nat Neurosci, 10, 321-330.
ZONOUZI M., SCAFIDI J., LI P., MCELLIN B., EDWARDS J., DUPREE J.L., HARVEY L., SUN D., HUBNER C.A., CULL-CANDY S.G., FARRANT M. & GALLO V. (2015) GABAergic regulation of cerebellar NG2 cell development is altered in perinatal white matter injury. Nat Neurosci, 18, 674-682.