Alzheimer’s disease (AD) is the most common cause of dementia with an increasing impact on the aging society. Although generations of researchers tried to unravel the pathomechanisms behind this disease, the molecular and cellular mechanisms leading to its onset and progression are still far from being completely understood. Accordingly, only a symptomatic treatment is available until now, and a curative treatment seems to be far-off. On the other hand, several novel therapeutic strategies have been proposed and debated during the last decade. Because of the extensive serotonergic denervation that has been observed in the AD brain and the important role played by serotonin in both, cognition and behavioural control, this neurotransmitter system has become a focus of a concerted research effort to identify new treatments for AD. Therefore, modulation of defined serotonin receptors by specific ligands represents a promising tool for treatments for neurodegenerative diseases like AD. Here we provide an overview of the involvement of the serotonergic system in AD and discuss the underlying molecular mechanisms.
Introduction
As the life expectancy increases and the population gets older, diseases of the elderly, like multiple forms of dementia, become one of the major concerns in Public Health worldwide and represent an increasing market for pharmaceutical companies. Alzheimer’s disease (AD) is the most common cause of severe memory loss and cognitive deterioration in the elderly, thus representing a major health concern in the ageing society. While there is no universal medical treatment available leading to a cure, a lot of effort has been made to identify potential targets for the development of novel drugs. Because of the extensive serotonergic denervation obtained in the AD brain, as well as an important role of serotonin (5-hydroxytryptamine; 5-HT) in both cognition and behavioural control, this neurotransmitter system has become a focus of a concerted research effort to identify new treatments for AD. Despite the importance of the serotonergic system in AD, we presently lack a complete understanding of the molecular downstream mechanisms underlying the effects of serotonin on AD. Herein, we will present the known details of the molecular signalling pathways of defined 5-HT receptors with respect to their possible role in AD onset and progression. We will also discuss the potential therapeutic approaches developed on the basis of this data.
Hallmarks of Alzheimer’s disease (AD)
An estimated 46.8 million people worldwide are living with dementia in the year 2015. This number will almost double every 20 years, reaching 74.7 million in 2030 and 131.5 million in the year 2050 (http://www.alz.co.uk/research/statistics). Hereby AD is the most common cause of dementia with a prevalence of 24 million and an incidence of up to 5 million per year (Ferri et al., 2005). Only in the USA, the estimated 5.3 million cases of AD account for direct costs to the American society of about 226 billion USD (http://www.alz.org/facts/). Accordingly, in the year 2050, where approximately 13.8 million people in USA will suffer from AD, the disease will produce costs of around 1.1 trillion USD. So the need for a medical breakthrough to cure the disease or at least slow the progression is urgent.
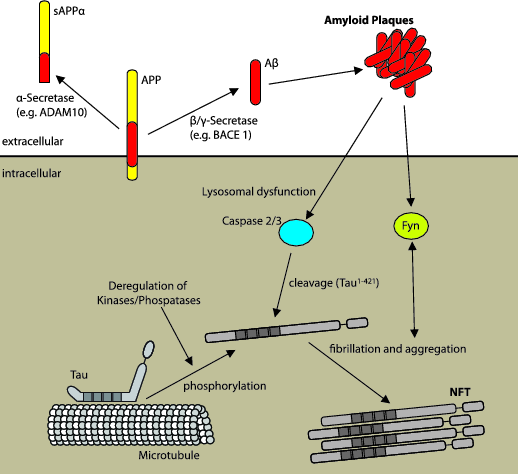
From the medical point of view, AD is characterized by an irreversible neurodegeneration, which slowly spreads over the whole brain and causes progressive memory loss, cognitive decline, and finally severe dementia. Although AD has been a topic of intensive research since it was first described 1906 by Alois Alzheimer (Alzheimer, 1906), the cellular and molecular mechanisms underlying it are not completely understood. Accordingly, an effective treatment or even cure except from moderate symptomatic relief has not been developed yet. As mentioned above, AD is diagnosed by a couple of different neurological symptoms, including loss of episodic memory, aphasia, apraxia and agnosia (Thomas and Fenech, 2006). However, the final diagnosis on a molecular base can only be done by the post-mortem screening for the main histopathological hallmarks of AD. These include two major types of aggregates (Fig. 1): (i) the extracellular amyloid plaques formed by insoluble aggregates of hydrophobic β-amyloid peptide (Aβ), representing fragments of the transmembrane amyloid Precursor Protein (APP) (Koo, 2002) and (ii) the intracellular fibrillary accumulations called Neurofibrillary tangles (NFTs) composed of hyper-phosphorylated tau protein (Microtubuli-associated protein tau, MAPT) (Iqbal et al., 1989, 2015).
Figure 1. Formation, processing and possible interplay between two main pathological hallmarks of AD. The pathological hallmarks of Alzheimer’s disease are extracellular Amyloid plaques and intracellular neurofibrillary tangles. Aβ, Amyloid protein fragment; APP, Amyloid Precursor Protein; sAPPα, soluble α-fragment of APP; NFT, Neurofibrillary tangles.
The formation of amyloid plaques results from the amyloidogenic degradation of the transmembrane precursor, the amyloid precursor protein (APP) by subsequent proteolytic cleavage with β-secretase (BACE-1) and γ-secretase. This leads to the formation of two amyloidogenic fragments of APP, Aβ40 and Aβ42. Amyloidogenic processing occurs mainly in early/sorting and late endosomes, where resulting Aβ peptides form oligomeric toxic species aggregating into extracellular senile plaques. An alternative non-amyloidogenic pathway is based on the cleavage of APP by an α-secretase (e.g. ADAM10). Cleavage at the α site located within the Aβ sequence prevents formation of the Aβ species and releases the extracellular soluble fragment of APP (sAPPα), which possesses neurotrophic and neuroprotective properties and can thus increase a long-term potentiation (Koo, 2002).
Tau can be abnormally hyper-phosphorylated by multiple proline-directed protein kinases (PDPKs). The main tau PDPKs are the glycogen synthase kinase‑3β (GSK‑3β) and the cyclin-dependent-like kinase 5 (CDK5). Although neither the levels nor the activities of these kinases are significantly altered in AD, truncation of GSK‑3β enhancing its activity has been found in the AD brain (Jin et al., 2015a, 2015b). However, the main regulator of tau phosphorylation is the protein phosphatase 2 (PPA2), the enzyme with the highest tau phosphatase activity in the human brain (Liu et al., 2005). PP2A regulates the phosphorylation of tau both directly as well as by regulating the activities of PDPKs. Dysregulation of the balanced activity of PDPKs and PPA2, followed by a hyper-phosphorylation of tau, leads to formation of paired helical filaments (PHFs) and NFTs.
Although enormous progress has been achieved in understanding the pathways leading to formation of aggregates, it still remains under intensive debate which species of the respective proteins and aggregates are responsible for the pathophysiology of AD. The story becomes even more complicated by multiple findings demonstrating that different pathological effects and symptoms might be induced by different toxic species. Therefore, it is very difficult to emphasize the importance of one hallmark over the other for the onset and progression of AD. Interestingly, although mutations correlated with an early-onset familial AD only appear in the genes coding for either APP or its processing proteins (e.g. Presenilin 1), they still lead to the formation of both, amyloid plaques as well as NFTs (Janssen et al., 2003). On the other hand, mutations in the gene coding for tau and inducing NFTs do not lead to the formation of amyloid plaques (Brandt et al., 2005). Such mutations do not induce AD, but lead to frontotemporal dementia with Parkinsonism linked to chromosome 17 (FTDP-17) (Foster et al., 1997; Pick, 1892). The first attempt to combine the two major hallmarks within a unifying theory was the amyloid cascade hypothesis formulated in 1992 by Hardy and Higgins (Hardy and Higgins, 1992). It explains the AD onset and progression as a consequence of downstream events induced by toxic species of the amyloid Aβ peptides. In this hypothesis, the formation of NFTs is described as a downstream event induced by Aβ accumulations which further contribute to intracellular toxicity (Busciglio et al., 1995). However, because the two hallmarks do appear in different spatial and temporal patterns within AD brains (Armstrong et al., 1993) and the severity of disease progression is rather correlated with the appearance of NFTs instead of Aβ plaques (Maccioni et al., 2001), the amyloid cascade hypothesis was relative quickly defeated. Later on, the revitalized tau hypothesis comprising the diverse findings and implying parallel pathways has been suggested by Maccioni and colleagues (Maccioni et al., 2010). According to this hypothesis, the cause of AD pathology is thought to be rather mediated by inflammatory conditions. Pro-inflammatory cytokines are able to dysregulate the activity of multiple kinases and phosphatases via different neuronal signaling cascades (Fernandez et al., 2008). As a consequence, tau protein becomes hyper-phosphorylated and forms PHFs, which manifest as NFTs in the neuronal soma. In addition, formation of the extracellular amyloid plaques might be a consequence of neuroinflammatory processes mediated by activated microglia (Maccioni et al., 2010), which can be triggered by tau filaments released from the dying cells. Activated microglia in turn will again produce inflammatory cytokines, which are described to activate several kinases like CDK5 (Alvarez et al., 2001; Maccioni et al., 2010). Thereby NFTs will also contribute to microglia activation due to dying neurons leading into a vicious cycle (Morales et al., 2010). In fact, this hypothesis suggests reciprocal connections between Aβ and tau as a cause of neuronal toxicity (Fig. 1). Accordingly, tau might also be involved in the triggering of Aβ toxicity. Tau is crucial for the post-synaptic localization of the src kinase Fyn responsible for phosphorylation of the NMDA receptor leading to excitotoxic downstream signaling (Ittner et al., 2010). On the other hand, caspase-2 and 3 are the downstream effectors of the toxic signaling mediated by the Aβ deposits, and activation of caspases leads to the formation of a truncated tau fragment (tau1-421), which is prone to accumulation (Gamblin et al., 2003). In addition, the lysosomes seem to play an important role since a malfunction of the lysosomes lead to an increase in tau1-421 and increased fibrillation and toxicity in a fly model (Butzlaff et al., 2015).
Serotonergic system and AD
The biogenic monoamine 5-HT acts as a tissue hormone, neurotransmitter and neuromodulator (Fidalgo et al., 2013; Ramakrishna et al., 2011). It was first isolated from enterochromaffin cells as a substance called enteramine (later named serotonin), inducing smooth muscle and vessel contraction in rat and mouse (Erspamer and Asero, 1952; Rapport et al., 1948a, 1948b; Vialli and Erspamer, 1937). Serotonin is one of the most extensively studied neurotransmitters in the central nervous system (CNS) regulating multiple physiological functions, including the control of anger, aggression, body temperature, appetite, sleep, mood and pain (Arreola et al., 2015; Mössner and Lesch, 1998). In the CNS, serotonin is produced by small clusters of serotonergic neurons located within the raphe area of the pons and the midbrain (B1–B9) (Dahlstroem and Fuxe, 1964). Serotonergic neurons comprise a widely distributed and complex network that targets nearly every brain structure.
Serotonin is the phylogenetically oldest neurotransmission system present in both, invertebrate and vertebrate species (Hay-Schmidt, 2000; Turlejski, 1996). It develops already within the first month of gestation in primates, long before other neurotransmitter systems arise (Levitt and Rakic, 1982). Neural serotonin concentration is increasing over the pre- and postnatal development with a peak one year after birth followed by a continuous decline (Azmitia and Whitaker-Azmitia, 2000), suggesting an important role of 5-HT in neuronal development and differentiation (Udo et al., 2005). In addition to its regulatory functions during development and neuronal differentiation, serotonin is critically involved in regulating learning processes and memory storage during adulthood and ageing. Therefore, pathological changes of the 5-HT metabolism and/or an imbalance in serotonergic signalling have been associated with the aetiology of various pathophysiological conditions in the CNS including AD, schizophrenia, depression, and anxiety (Bantick et al., 2001; Burnet et al., 1997; Lai et al., 2003; Weinstein et al., 1996). For example, it has been shown that AD is associated with a decreased number of serotonergic neurons in the raphe nuclei (Aletrino et al., 1992; Chen et al., 2000; Halliday et al., 1992; Kovacs et al., 2003; Yamamoto and Hirano, 1985; Zweig et al., 1988). In line with the decreased number of serotonergic neurons, the levels of 5-HT and its metabolites measured with high performance liquid chromatography (HPLC) in post-mortem AD brains is reduced (Garcia-Alloza et al., 2005; Gottfries, 1990). However, it is currently not clear whether atrophy of 5-HT neurons obtained in the brains of AD patients is a consequence of the general neurodegeneration or whether it represents a factor contributing to disease development and progression. It was shown in an APP transgenic mouse model of AD that amyloid depositions in the projection sites of serotonergic neurons lead to the degeneration of 5-HT axons followed by the degeneration of the corresponding neuronal cell bodies (Liu et al., 2008). On the other hand, several AD-associated abnormalities like hyper-phosphorylation of tau become primarily evident in the dorsal raphe nucleus, which subsequently leads to the 5-HT neuron demise (Hendricksen et al., 2004). The reduced levels of 5-HT within the projection regions might also contribute to AD progression. An importance of the 5-HT system in AD pathology is further confirmed by the observation, that neither 5-HT levels nor the number of serotonergic projections are affected in cases of Frontotemporal dementia (FTD), which exclusively shows tau hyperphosphorylation and NFT accumulation (Bowen et al., 2007).
Serotonin operates by activating a large family of specific 5-HT receptors (5-HTRs), which comprise seven distinct classes based on their structural and functional characteristics. Except the 5-HT3R, which is a transmitter-gated Na+/K+ channel, all other 5-HT receptors belong to the family of seven transmembrane domain receptors that are coupled to different intracellular effectors via heterotrimeric guanine nucleotide binding proteins (G-proteins) (Barnes and Sharp, 1999). The 5-HT1R is coupled to inhibitory Gi-proteins leading to an inhibition of adenylyl cyclases (AC). The three isoforms of 5-HT2Rs (5-HT2A, 5-HT2B and 5-HT2C) are coupled to heterotrimeric Gq-proteins that are linked to the phospholipase-C (PLC) signaling pathway. The effector system for the two isoforms of 5-HT5Rs, 5-HT5AR and 5-HT5BR, remains to be clarified, although there is information that the 5-HT5AR is coupled to the PTX-sensitive Gi/0 proteins and AC (Francken et al., 1998; Hurley et al., 1998). Other 5-HTR subfamilies are 5-HT4R, 5-HT6R and 5-HT7R, which are linked to Gs-proteins leading to activation of ACs. The 5-HT4R and 5-HT7R can also activate heterotrimeric G12/13 proteins, which in turn selectively stimulate small GTPases of the RhoA family resulting in pronounced morphological changes at the cellular level (Kobe et al., 2012; Kvachnina, 2005; Ponimaskin et al., 2007).
Serotonin receptors and amyloid beta plaques
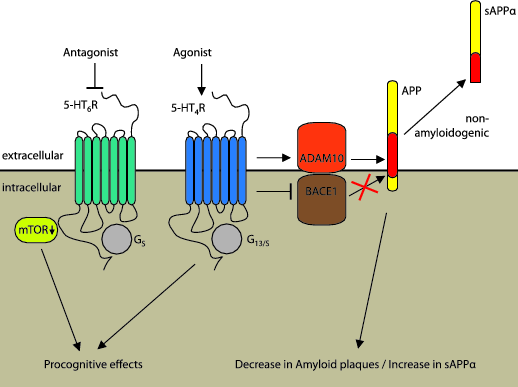
Most of the publications dealing with the role of serotonin receptors in AD focus on the possible interplay between the serotonergic system and the amyloid-mediated part of pathophysiology. This is mostly based on the experimental finding demonstrating that administration of selective serotonin reuptake inhibitors (SSRI) to mouse models of AD reduces the production of toxic amyloid proteins and amyloid plaques (Cirrito et al., 2011; Sheline et al., 2014). SSRIs are targeting 5-HT transporter proteins in brain, with the aim of increasing serotonergic neurotransmission. They are the current first line drugs used as long-term antidepressant treatments and show only minor side effects. The fact that SSRIs seem to inhibit the production of toxic amyloid species makes them a promising tool to slow-down the progression of AD (Claeysen et al., 2015). The observation that modulating the serotonergic system may have a beneficial effect on the AD treatment initiated multiple studies to investigate which serotonin receptor(s) might be involved. Several 5-HT receptors have been shown to influence processing of the amyloid protein precursor (APP), including 5-HT2AR, 5-HT2CR, and 5-HT4R (Cochet et al., 2013; Thathiah and De Strooper, 2011). Among them, the 5-HT4R was identified as the most promising target, since activation of this receptor induces the non-amyloidogenic cleavage of APP, which results in a release of the soluble sAPPα fragment possessing neurotrophic and neuroprotective properties (Cochet et al., 2013). In the recent study, Cochet and co-workers demonstrated that chronic stimulation of the 5-HT4R with a partial agonist RS 67333 slowed down the production of Aβ plaques and prevented cognitive deficits in a 5XFAD mouse model of early onset AD by promoting the α-secretase cleavage of APP (Cochet et al., 2013). The mechanism behind implies a direct interaction of the 5-HT4R with a major alpha-secretase of APP: ADAM10, which in turn induces the non-amyloidogenic pathway of APP cleavage (Fig. 2). In addition, chronic administration of RS 67333 resulted in significantly reduced cerebral inflammation processes (i.e. astrogliosis and microgliosis), which is associated with AD progression. At the cellular level, anti-inflammatory effects can be explained by the 5-HT4R-mediated rise in cAMP production, which is known to have an anti-inflammatory action (Cai et al., 1999). Moreover, acute administration of RS 67333 led to a transient increase in concentration of sAPPα in the cerebrospinal fluid of a mouse model of AD (Cochet et al., 2013). In another study published in parallel, Tesseur and colleagues found that a chronic administration of the novel 5-HT4R agonist SSP-002392 in a hAPP/PS1 mouse model of AD decreased the amount of both, soluble as well as insoluble Aβ in the brain (Tesseur et al., 2013). The authors also demonstrated an increased astro- and microgliosis surrounding the Aβ deposits in the brain. This finding is in line with previous publications showing an immunomodulatory role of 5-HT4R (Müller et al., 2009; Zeinstra et al., 2006). Interestingly, the authors did not find any evidence for direct α-secretase stimulation in the brain, but rather decreased BACE-1 expression upon chronic treament with SSP-002392 (Tesseur et al., 2013). BACE-1 is the rate-limiting enzyme of Aβ production, and BACE-1 deficiency is known to decrease Aβ levels, as well as amyloid plaque deposition in the brain of an AD mouse model (McConlogue et al., 2007). Based on these observations, the authors concluded that the reduction of BACE-1 and the increased micro- and astrogliosis could represent the molecular mechanisms responsible for the observed decrease in Aβ.
Figure 2. Serotonin receptors and APP processing. The impact of 5-HT4R and 5-HT6R-mediated signaling on the APP degradation. While stimulation of the 5-HT4R drives APP processing towards the beneficial non-amyloidogenic path via direct interaction with α-secretase ADAM10 or β-secretase BACE1, inhibition of the 5-HT6 receptor promotes procognitive effects via reduction of mTOR activity.
Another 5-HT receptor that has recently received attention as a potential target for the AD treatment is the 5-HT6R. Although, similarly to 5-HT4R, this receptor is coupled to the Gs-protein to stimulate AC, several reports suggest that beneficial effects on cognition arise from 5-HT6R inactivation. Administration of 5-HT6R antagonists to rodent models of AD improves cognitive performance in numerous behavioral tests (Upton et al., 2008). More recently, results obtained in the phase II clinical trial for 5-HT6R antagonist idalopirdine demonstrated cognitive improvement in patients with moderate AD who received idalopirdine as a combination therapy with an inhibitor of Acetylcholinesterase donepezil, which is routinely used for a symptomatic AD treatment (Wilkinson et al., 2014). At the cellular level, beneficial effects of 5-HT6R inhibition might be evoked due to inhibition of the mTOR pathway, rather than Gs-mediated modulation of proteases involved in Aβ processing (e.g. ADAM10) (Wang et al., 2013). However, detailed molecular mechanisms still remain elusive.
Serotonin receptors and neurofibrillary tangles (NFTs)
Hyper-phosphorylaton of tau is mainly mediated by PDPKs including GSK-3β (Arioka et al., 1993), CDK5 (Imahori and Uchida, 1997; Ishiguro et al., 1992) and the dual specificity tyrosine-phosphorylation-regulated kinase 1A (DYRK1A) (Woods et al., 2001). In addition, there are several non-PDPKs known to phosphorylate tau, such as calcium/calmodulin-activated protein kinase II (CaMKII) (Baudier and Cole, 1988), microtubule affinity-regulated kinase 110 (MARK p110) (Ledesma et al., 1992), protein kinase A (PKA) (Singh et al., 1996a), casein kinase 1 (CK1) (Singh et al., 1996b) and the src family tyrosine kinase Fyn (Lee et al., 2004). However, the non-PDPKs seems to only prime the tau hyper-phosphorylation by the three PDPKs via single phosphorylation (Liu et al., 2006; Singh et al., 1995a, 1995b). The regulation of phosphorylation is mediated by PP2A, which is found to be impaired in AD (Gong et al., 1993). Noteworthy, PP2A is not only regulating the tau phosphorylation directly, but is also known to regulate kinase activity of GSK-3β, CDK5, CaMKII and PKA (Iqbal et al., 2005), suggesting that PP2A might represent an important pharmacological target (Iqbal et al., 2015; Sontag and Sontag, 2014). Interestingly, PPA2 can also be involved in regulating the concentration of biogenic amines, including serotonin. For example, a regulated association of serotonin transporter (SERT) with the catalytic subunit of PP2A has been demonstrated in both a recombinant system as well as in the brain tissue (Bauman et al., 2000). From the functional point of view, SERT-associated PP2A may control the phosphorylation state of one or more transporter-associated proteins that influence transporter intrinsic activity and cytoskeletal associations. Moreover, because transporter phosphorylation is selectively elevated by PP2A inhibitors (Ramamoorthy et al., 1998), PP2A may target phosphorylation sites on SERT itself (Qian et al., 1995). Thus, impaired functional activity of PP2A obtained in AD can also affect the physiological serotonin level in different brain areas, which might be a reason for multiple neuropsychiatric symptoms (i.e. depression) often observed in AD patients.
Figure 2. Serotonin receptors and APP processing. The impact of 5-HT4R and 5-HT6R-mediated signaling on the APP degradation. While stimulation of the 5-HT4R drives APP processing towards the beneficial non-amyloidogenic path via direct interaction with α-secretase ADAM10 or β-secretase BACE1, inhibition of the 5-HT6 receptor promotes procognitive effects via reduction of mTOR activity.
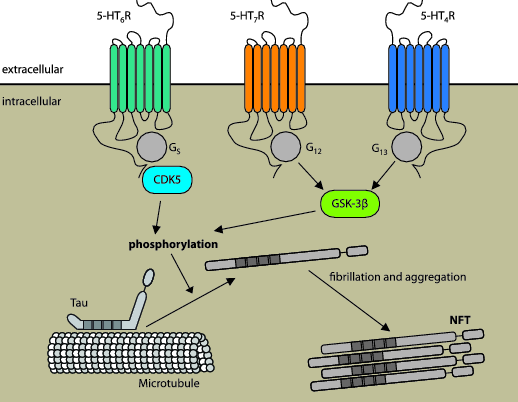
Whereas serotonin receptors (e.g. 5-HT4R and 5-HT6R) can influence processing of Aβ via modulation of activity of corresponding secretases (e.g ADAM10 or BACE1), the role of serotonin receptors in NFT formation have not yet been analysed. However, there is indirect evidence suggesting the involvement of the serotonergic system in tau hyper-phosphorylation. For example, Yun and coworkers have demonstrated the physical and functional association of 5-HT6R with Fyn kinase, which suggest an important role on 5-HT6R in modulation of tau hyper-phosphorylation (Yun et al., 2007). In this study, the authors found that activation of 5-HT6R results in phosphorylation-dependent activation of Fyn as well as Fyn-dependent activation of the extracellular-signal regulated kinases 1 and 2 (ERK1/2). This finding could have an important implication in AD, because of the potentially negative effects of active Fyn on tau phosphorylation (compare Fig. 1) (Lee et al., 2004). It can thus be speculated that 5-HT6R-mediated activation of Fyn would lead to pathological tau hyper-phosphorylation, while the 5-HT6R inhibition may be protective. In addition to the modulation of non-PDPK activity, 5-HT6R can directly interact with and activate CDK5, one of the major tau PDPKs (Duhr et al., 2014). However, in this study, the authors did not analyze the implications of 5-HT6R-induced CDK5 activation on tau pathology. Therefore, future investigations will be necessary to more precisely evaluate the role of the 5-HT6R/CDK5 interaction for NFT formation.
Another putative connection between serotonin receptors and tau phosphorylation could be mediated by GSK-3β. It has been shown that in neuronal cells GSK-3β can be activated by both Gα12 and Gα13 subunits (Sayas et al., 2002). Interestingly, whereas Gα13-mediated GSK-3β activation was Rho-mediated, GSK-3β activation by Gα12 is Rho-independent. These findings mean that serotonin receptors coupled to G12/13 proteins might potentially contribute to tau phosphorylation and pathology via modulation of GSK-3β activity. (Fig. 3). We have previously demonstrated that the 5-HT4R is coupled to Gα13 protein and that stimulation of 5-HT4R leads to Gα13-mediated activation of the small GTPase RhoA (Pindon et al., 2002; Ponimaskin et al., 2002). In addition, we have found that 5-HT7R is coupled to the Gα12 subunit. A detailed analysis of 5-HT7R-mediated signalling reveals that 5-HT7R/G12 signalling activates both RhoA and Cdc42 (Kobe et al., 2012; Kvachnina, 2005). Thus, by activating G12/13 proteins both 5-HT4 and 5-HT7 receptors could also be potential activators of GSK-3β (Fig. 3).
An additional and even more intrigue connection between tau pathology and 5-HT receptors involves the receptor-mediated modulation of the Brain-derived neurotrophic factor (BDNF) concentration. It is known that BDNF attenuates tau and Aβ pathologies by activating the phosphatidylinositol 3 kinase/GSK-3β pathway. Activation of this pathway, which leads to the inhibition of GSK-3β activity, attenuates phosphorylation of tau and, consequently, reduces pathology (Kazim et al., 2014). Also it is known that stimulation of 5-HT2AR results in increased BDNF expression in the neocortex and hippocampus of rats (Vaidya et al., 1997). Also the activation of 5-HT4R leads to increased levels of BDNF via the CREB-mediated pathway. Thus, modulation of BDNF concentration via 5-HT2AR and 5-HT4R might represent a novel target in AD treatment.
Conclusion
The serotonergic system, although quite complex, represents a very potent therapeutic target for the treatment of AD. Such kind of therapy can gain relative high specificity because for the most serotonin receptor subtypes selective agonists as well as antagonists are presently available. As long as molecular mechanisms underlying the impact of serotoninergic signaling in AD are not completely understood, general treatment approaches using SSRIs might also be of interest (Cirrito et al., 2011; Sheline et al., 2014). Still, non-specific activation of multiple serotonin receptors achieved by SSRIs can result in opposing effects. Indeed, while activation of 5-HT4R, 5-HT2AR and 5-HT7R might be beneficial, stimulation of the 5-HT6R might deteriorate AD symptoms. Thus, combined application of agonists and antagonist for defined serotonin receptors may result in higher therapeutic benefit. For that, the impact of single 5-HT receptors on the pathomechanisms of AD or related dementias has to be elucidated in more detail.
| Attachment | Size |
|---|---|
| 454.85 KB |
Financial support was provided by the BMBF (Federal Ministry of Education and Research, Germany, Grant 0315690D), the Deutsche Forschungsgemeinschaft (DFG, German Research Foundation, Grant PO732), the BMBF and Neuron EraNet (project TargetECM, 01EW1308B) and the Deutsche Forschungsgemeinschaft for the Cluster of Excellence REBIRTH (EXC 62/3). The funders had no role in the study design, data collection and analysis, decision to publish, or preparation of the manuscript.
ALETRINO M.A., VOGELS O.J.M., VAN DOMBURG P.H.M.F., TEN DONKELAAR H.J. (1992). Cell loss in the nucleus raphes dorsalis in alzheimer’s disease. Neurobiol. Aging 13, 461–468.
ALVAREZ A., MUÑOZ J.P., MACCIONI R.B. (2001). A Cdk5–p35 Stable Complex Is Involved in the β-Amyloid-Induced Deregulation of Cdk5 Activity in Hippocampal Neurons. Exp. Cell Res. 264, 266–274.
ALZHEIMER A. (1906). Über einen eigenartigen schweren Erkrankungsprozeβ der Hirnrincde. Neurol. Cent. 1129–1136.
ARIOKA M., TSUKAMOTO M., ISHIGURO K., KATO R., SATO K., IMAHORI K., UCHIDA T. (1993). τ Protein Kinase II Is Involved in the Regulation of the Normal Phosphorylation State of τ Protein. J. Neurochem. 60, 461–468.
ARMSTRONG R.A., MYERS D., SMITH C.U. (1993). The spatial patterns of plaques and tangles in Alzheimer’s disease do not support the “cascade hypothesis.” Dementia 4, 16–20.
ARREOLA R., BECERRIL-VILLANUEVA E., CRUZ-FUENTES C., VELASCO-VELÁZQUEZ M.A., GARCÉS-ALVAREZ M.E., HURTADO-ALVARADO G., QUINTERO-FABIAN S., PAVÓN L. (2015). Immunomodulatory Effects Mediated by Serotonin. J. Immunol. Res. 2015.
AZMITIA E.C., WHITAKER-AZMITIA P.M. (2000). Development and Adult Plasticity of Serotoninergic Neurons and Their Target Cells. In Serotoninergic Neurons and 5-HT Receptors in the CNS, P.D. med H.G.B. M.D, and P.D. med M. Göthert, eds. (Springer Berlin Heidelberg), pp. 1–39.
BANTICK R.A., DEAKIN J.F., GRASBY P.M. (2001). The 5-HT1A receptor in schizophrenia: a promising target for novel atypical neuroleptics? J. Psychopharmacol. Oxf. Engl. 15, 37–46.
BAUDIER J., COLE R.D. (1988). Interactions between the microtubule-associated tau proteins and S100b regulate tau phosphorylation by the Ca2+/calmodulin-dependent protein kinase II. J. Biol. Chem. 263, 5876–5883.
BAUMAN A.L., APPARSUNDARAM S., RAMAMOORTHY S., WADZINSKI B.E., VAUGHAN R.A., BLAKELY R.D. (2000). Cocaine and antidepressant-sensitive biogenic amine transporters exist in regulated complexes with protein phosphatase 2A. J. Neurosci. Off. J. Soc. Neurosci. 20, 7571–7578.
BOWEN D.M., PROCTER A.W., MANN D.M.A., SNOWDEN J.S., ESIRI M.M., NEARY D., FRANCIS P.T. (2007). Imbalance of a serotonergic system in frontotemporal dementia: implication for pharmacotherapy. Psychopharmacology (Berl.) 196, 603–610.
BRANDT R., HUNDELT M., SHAHANI N. (2005). Tau alteration and neuronal degeneration in tauopathies: mechanisms and models. Biochim Biophys Acta 1739, 331–354.
BURNET P.W., EASTWOOD S.L., HARRISON P.J. (1997). [3H]WAY-100635 for 5-HT1A receptor autoradiography in human brain: a comparison with [3H]8-OH-DPAT and demonstration of increased binding in the frontal cortex in schizophrenia. Neurochem. Int. 30, 565–574.
BUSCIGLIO J., LORENZO A., YEH J., YANKNER B.A. (1995). beta-amyloid fibrils induce tau phosphorylation and loss of microtubule binding. Neuron 14, 879–888.
BUTZLAFF M., HANNAN S.B., KARSTEN P., LENZ S., NG J., VOßFELDT H., PRÜßING K., PFLANZ R., SCHULZ J.B., RASSE T., Et Al. (2015). Impaired retrograde transport by the Dynein/Dynactin complex contributes to tau-induced toxicity. Hum. Mol. Genet. 24, 3623–3637.
CAI D., SHEN Y., DE BELLARD M., TANG S., FILBIN M.T. (1999). Prior Exposure to Neurotrophins Blocks Inhibition of Axonal Regeneration by MAG and Myelin via a cAMP-Dependent Mechanism. Neuron 22, 89–101.
CHEN C.P.L.-H., EASTWOOD S.L., HOPE T., MCDONALD B., FRANCIS P.T., ESIRI M.M. (2000). Immunocytochemical study of the dorsal and median raphe nuclei in patients with Alzheimer’s disease prospectively assessed for behavioural changes. Neuropathol. Appl. Neurobiol. 26, 347–355.
CIRRITO J.R., DISABATO B.M., RESTIVO J.L., VERGES D.K., GOEBEL W.D., SATHYAN A., HAYREH D., D’ANGELO G., BENZINGER T., YOON H., Et Al. (2011). Serotonin signaling is associated with lower amyloid-β levels and plaques in transgenic mice and humans. Proc. Natl. Acad. Sci. 108, 14968–14973.
CLAEYSEN S., BOCKAERT J., GIANNONI P. (2015). Serotonin: A New Hope in Alzheimer’s Disease? ACS Chem. Neurosci. 6, 940–943.
COCHET M., DONNEGER R., CASSIER E., GAVEN F., LICHTENTHALER S.F., MARIN P., BOCKAERT J., DUMUIS A., CLAEYSEN S. (2013). 5-HT4 Receptors Constitutively Promote the Non-Amyloidogenic Pathway of APP Cleavage and Interact with ADAM10. ACS Chem. Neurosci. 4, 130–140.
DAHLSTROEM A., FUXE K. (1964). Evidence for the existence of monoamine-containing neurons in the central nervous system. I. Demonstration of monoamines in the cell bodies of brain stem neurons. Acta Physiol. Scand. Suppl. SUPPL 232:1–55.
DUHR F., DÉLÉRIS P., RAYNAUD F., SÉVENO M., MORISSET-LOPEZ S., MANNOURY LA COUR C., MILLAN M.J., BOCKAERT J., MARIN P., CHAUMONT-DUBEL S. (2014). Cdk5 induces constitutive activation of 5-HT6 receptors to promote neurite growth. Nat. Chem. Biol. 10, 590–597.
ERSPAMER V., ASERO B. (1952). Identification of Enteramine, the Specific Hormone of the Enterochromaffin Cell System, as 5-Hydroxytryptamine. Nature 169, 800–801.
FERNANDEZ J.A., ROJO L., KULJIS R.O., MACCIONI R.B. (2008). The damage signals hypothesis of Alzheimer’s disease pathogenesis. J Alzheimers Dis 14, 329–333.
FERRI C.P., PRINCE M., BRAYNE C., BRODATY H., FRATIGLIONI L., GANGULI M., HALL K., HASEGAWA K., HENDRIE H., HUANG Y., Et Al. (2005). Global prevalence of dementia: a Delphi consensus study. Lancet 366, 2112–2117.
FIDALGO S., IVANOV D.K., WOOD S.H. (2013). Serotonin: from top to bottom. Biogerontology 14, 21–45.
FOSTER N.L., WILHELMSEN K., SIMA A.A., JONES M.Z., D’AMATO C.J., GILMAN S. (1997). Frontotemporal dementia and parkinsonism linked to chromosome 17: a consensus conference. Conference Participants. Ann Neurol 41, 706–715.
FRANCKEN B.J., JURZAK M., VANHAUWE J.F., LUYTEN W.H., LEYSEN J.E. (1998). The human 5-ht5A receptor couples to Gi/Go proteins and inhibits adenylate cyclase in HEK 293 cells. Eur. J. Pharmacol. 361, 299–309.
GAMBLIN T.C., CHEN F., ZAMBRANO A., ABRAHA A., LAGALWAR S., GUILLOZET A.L., LU M., FU Y., GARCIA-SIERRA F., LAPOINTE N., Et Al. (2003). Caspase cleavage of tau: linking amyloid and neurofibrillary tangles in Alzheimer’s disease. Proc. Natl. Acad. Sci. 100, 10032–10037.
GARCIA-ALLOZA M., GIL-BEA F.J., DIEZ-ARIZA M., CHEN C.P.L.-H., FRANCIS P.T., LASHERAS B., RAMIREZ M.J. (2005). Cholinergic–serotonergic imbalance contributes to cognitive and behavioral symptoms in Alzheimer’s disease. Neuropsychologia 43, 442–449.
GONG C.-X., SINGH T.J., GRUNDKE-IQBAL I., IQBAL K. (1993). Phosphoprotein Phosphatase Activities in Alzheimer Disease Brain. J. Neurochem. 61, 921–927.
GOTTFRIES P.D.C.G. (1990). Disturbance of the 5-hydroxytryptamine metabolism in brains from patients with Alzheimer’s dementia. In Neurotransmitter and Dementia, P.D.C.G. Gottfries, and P.D.S. Nakamura, eds. (Springer Vienna), pp. 33–43.
HALLIDAY G.M., MCCANN H.L., PAMPHLETT R., BROOKS W.S., CREASEY H., MCCUSKER E., COTTON R.G.H., BROE G.A., HARPER C.G. (1992). Brain stem serotonin-synthesizing neurons in Alzheimer’s disease: a clinicopathological correlation. Acta Neuropathol. (Berl.) 84, 638–650.
HARDY J.A., HIGGINS G.A. (1992). Alzheimer’s disease: the amyloid cascade hypothesis. Science 256, 184–185.
HAY-SCHMIDT A. (2000). The evolution of the serotonergic nervous system. Proc. R. Soc. B Biol. Sci. 267, 1071–1079.
HENDRICKSEN M., THOMAS A.J., FERRIER I.N., INCE P., O’BRIEN J.T. (2004). Neuropathological Study of the Dorsal Raphe Nuclei in Late-Life Depression and Alzheimer’s Disease With and Without Depression. Am. J. Psychiatry 161, 1096–1102.
HURLEY P.T., MCMAHON R.A., FANNING P., O’BOYLE K.M., ROGERS M., MARTIN F. (1998). Functional coupling of a recombinant Human 5-HT5A receptor to G-proteins in HEK-293 cells. Br. J. Pharmacol. 124, 1238–1244.
IMAHORI K., UCHIDA T. (1997). Physiology and pathology of tau protein kinases in relation to Alzheimer’s disease. J. Biochem. (Tokyo) 121, 179–188.
IQBAL K., GRUNDKE-IQBAL I., SMITH A.J., GEORGE L., TUNG Y.C., ZAIDI T. (1989). Identification and localization of a tau peptide to paired helical filaments of Alzheimer disease. Proc Natl Acad Sci U A 86, 5646–5650.
IQBAL K., DEL C. ALONSO A., CHEN S., CHOHAN M.O., EL-AKKAD E., GONG C.-X., KHATOON S., LI B., LIU F., RAHMAN A., Et Al. (2005). Tau pathology in Alzheimer disease and other tauopathies. Biochim. Biophys. Acta BBA - Mol. Basis Dis. 1739, 198–210.
IQBAL K., LIU F., GONG C.-X. (2015). Tau and neurodegenerative disease: the story so far. Nat. Rev. Neurol.
ISHIGURO K., TAKAMATSU M., TOMIZAWA K., OMORI A., TAKAHASHI M., ARIOKA M., UCHIDA T., IMAHORI K. (1992). Tau protein kinase I converts normal tau protein into A68-like component of paired helical filaments. J. Biol. Chem. 267, 10897–10901.
ITTNER L.M., KE Y.D., DELERUE F., BI M., GLADBACH A., VAN EERSEL J., WÖLFING H., CHIENG B.C., CHRISTIE M.J., NAPIER I.A., Et Al. (2010). Dendritic Function of tau Mediates Amyloid-β Toxicity in Alzheimer’s Disease Mouse Models. Cell 142, 387–397.
JANSSEN J.C., BECK J.A., CAMPBELL T.A., DICKINSON A., FOX N.C., HARVEY R.J., HOULDEN H., ROSSOR M.N., COLLINGE J. (2003). Early onset familial Alzheimer’s disease: Mutation frequency in 31 families. Neurology 60, 235–239.
JIN N., YIN X., GU J., ZHANG X., SHI J., QIAN W., JI Y., CAO M., GU X., DING F., Et Al. (2015a). Truncation and Activation of Dual Specificity Tyrosine Phosphorylation-regulated Kinase 1A by Calpain I: A Molecular Mechanism linked to tau Pathology in Alzheimer Disease. J. Biol. Chem. 290, 15219–15237.
JIN N., YIN X., YU D., CAO M., GONG C.-X., IQBAL K., DING F., GU X., LIU, F. (2015b). Truncation and activation of GSK-3β by calpain I: a molecular mechanism links to tau hyperphosphorylation in Alzheimer’s disease. Sci. Rep. 5, 8187.
KAZIM S.F., BLANCHARD J., DAI C.-L., TUNG Y.-C., LAFERLA F.M., IQBAL I.-G., IQBAL K. (2014). Disease modifying effect of chronic oral treatment with a neurotrophic peptidergic compound in a triple transgenic mouse model of Alzheimer’s disease. Neurobiol. Dis. 71, 110–130.
KOBE F., GUSEVA D., JENSEN T.P., WIRTH A., RENNER U., HESS D., MULLER M., MEDRIHAN L., ZHANG W., ZHANG M. Et Al. (2012). 5-HT7R/G12 Signaling Regulates Neuronal Morphology and Function in an Age-Dependent Manner. J. Neurosci. Off. J. Soc. Neurosci. 32, 2915–2930.
KOO E.H. (2002). The beta-amyloid precursor protein (APP) and Alzheimer’s disease: does the tail wag the dog? Traffic 3, 763–770.
KOVACS G.G., KLÖPPEL S., FISCHER I., DORNER S., LINDECK-POZZA E., BIRNER P., BÖTEFÜR I.C., PILZ P., VOLK B., BUDKA H. (2003). Nucleus-specific alteration of raphe neurons in human neurodegenerative disorders. Neuroreport 14, 73–76.
KVACHNINA E. (2005). 5-HT7 Receptor Is Coupled to G Subunits of Heterotrimeric G12-Protein to Regulate Gene Transcription and Neuronal Morphology. J. Neurosci. 25, 7821–7830.
LAI M.K.P., TSANG S.W.Y., FRANCIS P.T., ESIRI M.M., KEENE J., HOPE T., CHEN C.P.L.-H. (2003). Reduced serotonin 5-HT1A receptor binding in the temporal cortex correlates with aggressive behavior in Alzheimer disease. Brain Res. 974, 82–87.
LEDESMA M.D., CORREAS I., AVILA J., DIAZ-NIDOJ. (1992). Implication of brain cdc2 and MAP2 kinases in the phosphorylation of tau protein in Alzheimer’s disease. FEBS Lett. 308, 218–224.
LEE G., THANGAVEL R., SHARMA V.M., LITERSKY J.M., BHASKAR K., FANG S.M., DO L.H., ANDREADIS A., HOESEN G.V., KSIEZAK-REDINGH. (2004). Phosphorylation of tau by Fyn: Implications for Alzheimer’s Disease. J. Neurosci. 24, 2304–2312.
LEVITT P., RAKIC P. (1982). The time of genesis, embryonic origin and differentiation of the brain stem monoamine neurons in the rhesus monkey. Brain Res. 256, 35–57.
LIU F., GRUNDKE-IQBAL I., IQBAL K., GONG C.-X. (2005). Contributions of protein phosphatases PP1, PP2A, PP2B and PP5 to the regulation of tau phosphorylation. Eur. J. Neurosci. 22, 1942–1950.
LIU F., LIANG Z., SHI J., YIN D., EL-AKKAD E., GRUNDKE-IQBAL I., IQBAL K., GONG C.-X. (2006). PKA modulates GSK-3beta- and cdk5-catalyzed phosphorylation of tau in site- and kinase-specific manners. FEBS Lett. 580, 6269–6274.
LIU Y., YOO M.-J., SAVONENKO A., STIRLING W., PRICE D.L., BORCHELT D.R., MAMOUNAS L., LYONS W.E., BLUE M.E., LEE M.K. (2008). Amyloid Pathology Is Associated with Progressive Monoaminergic Neurodegeneration in a Transgenic Mouse Model of Alzheimer’s Disease. J. Neurosci. 28, 13805–13814.
MACCIONI R.B., MUNOZ J.P., BARBEITO L. (2001). The molecular bases of Alzheimer’s disease and other neurodegenerative disorders. Arch Med Res 32, 367–381.
MACCIONI R.B., FARÍAS G., MORALES I., NAVARRETE L. (2010). The Revitalized Tau Hypothesis on Alzheimer’s Disease. Arch. Med. Res. 41, 226–231.
MCCONLOGUE L., BUTTINI M., ANDERSON J.P., BRIGHAM E.F., CHEN K.S., FREEDMAN S.B., GAMES D., JOHNSON-WOOD K., LEE M., ZELLER M., Et Al. (2007). Partial reduction of BACE1 has dramatic effects on Alzheimer plaque and synaptic pathology in APP Transgenic Mice. J. Biol. Chem. 282, 26326–26334.
MORALES I., FARIAS G., MACCIONI R.B. (2010). Neuroimmunomodulation in the pathogenesis of Alzheimer’s disease. Neuroimmunomodulation 17, 202–204.
MÖSSNER R., LESCH K.-P. (1998). Role of Serotonin in the Immune System and in Neuroimmune Interactions. Brain. Behav. Immun. 12, 249–271.
MÜLLER T., DÜRK T., BLUMENTHAL B., GRIMM M., CICKO S., PANTHER E., SORICHTER S., HEROUY Y., DI VIRGILIO F., FERRARI D. Et Al. (2009). 5-Hydroxytryptamine Modulates Migration, Cytokine and Chemokine Release and T-Cell Priming Capacity of Dendritic Cells In Vitro and In Vivo. PLoS ONE 4, e6453.
PICK A. (1892). Über die Beziehungen der senilen Hirnatrophie zur Aphasie. Prag. Med. Wochenschr. 17, 165–167.
PINDON A., VAN HECKE G., VAN GOMPEL P., LESAGE A.S., LEYSEN J.E., JURZAK M. (2002). Differences in signal transduction of two 5-HT4 receptor splice variants: compound specificity and dual coupling with Galphas- and Galphai/o-proteins. Mol. Pharmacol. 61, 85–96.
PONIMASKIN E., VOYNO-YASENETSKAYA T., RICHTER D.W., SCHACHNER M., DITYATEV A. (2007). Morphogenic Signaling in Neurons Via Neurotransmitter Receptors and Small GTPases. Mol. Neurobiol. 35, 278–287.
PONIMASKIN E.G., PROFIROVIC J., VAISKUNAITE R., RICHTER D.W., VOYNO-YASENETSKAYA T.A. (2002). 5-Hydroxytryptamine 4(a) Receptor Is Coupled to the G Subunit of Heterotrimeric G13 Protein. J. Biol. Chem. 277, 20812–20819.
QIAN Y., MELIKIAN H., MOORE K., DUKE B., BLAKELY R. (1995). Phosphorylation of serotonin transporter domains and their role of phosphorylation in acute transporter regulation. In Soc Neurosci Abstr,.
RAMAKRISHNA A., GIRIDHAR P., RAVISHANKAR G.A. (2011). Phytoserotonin. Plant Signal. Behav. 6, 800–809.
RAMAMOORTHY S., GIOVANETTI E., QIAN Y., BLAKELY R.D. (1998). Phosphorylation and Regulation of Antidepressant-sensitive Serotonin Transporters. J. Biol. Chem. 273, 2458–2466.
RAPPORT M.M., GREEN A.A., PAGE I.H. (1948a). Serum vasoconstrictor, serotonin; isolation and characterization. J. Biol. Chem. 176, 1243–1251.
RAPPORT M.M., GREEN A.A., PAGE I.H. (1948b). Partial Purification of the Vasoconstrictor in Beef Serum. J. Biol. Chem. 174, 735–741.
SAYAS C.L., AVILA J., WANDOSELL F. (2002). Glycogen synthase kinase-3 is activated in neuronal cells by Gα12 and Gα13 by Rho-independent and Rho-dependent mechanisms. J. Neurosci. 22, 6863–6875.
SHELINE Y.I., WEST T., YARASHESKI K., SWARM R., JASIELEC M.S., FISHER J.R., FICKER W.D., YAN P., XIONG C., FREDERIKSEN C. Et Al. (2014). An Antidepressant Decreases CSF Aβ Production in Healthy Individuals and in Transgenic AD Mice. Sci. Transl. Med. 6, 236re4–re236re4.
SINGH T.J., HAQUE N., GRUNDKE-IQBAL I., IQBAL K. (1995a). Rapid Alzheimer-like phosphorylation of tau by the synergistic actions of non-proline-dependent protein kinases and GSK-3. FEBS Lett. 358, 267–272.
SINGH T.J., ZAIDI T., GRUNDKE-IQBAL I., IQBAL K. (1995b). Modulation of GSK-3-catalyzed phosphorylation of microtubule-associated protein tau by non-proline-dependent protein kinases. FEBS Lett. 358, 4–8.
SINGH T.J., GRUNDKE-IQBAL I., IQBAL K. (1996a). Differential phosphorylation of human tau isoforms containing three repeats by several protein kinases. Arch. Biochem. Biophys. 328, 43–50.
SINGH T.J., ZAIDI T., GRUNDKE-IQBAL I., IQBAL K. (1996b). Non-proline-dependent protein kinases phosphorylate several sites found in tau from Alzheimer disease brain. Mol. Cell. Biochem. 154, 143–151.
SONTAG J.-M., SONTAG E. (2014). Protein phosphatase 2A dysfunction in Alzheimer’s disease. Front. Mol. Neurosci. 7.
TESSEUR I., PIMENOVA A.A., LO A.C., CIESIELSKA M., LICHTENTHALER S.F., DE MAEYER J.H., SCHUURKES J.A.J., D’HOOGE R., DE STROOPER B. (2013). Chronic 5-HT4 receptor activation decreases Aβ production and deposition in hAPP/PS1 mice. Neurobiol. Aging 34, 1779–1789.
THATHIAH A., DE STROOPER B. (2011). The role of G protein-coupled receptors in the pathology of Alzheimer’s disease. Nat. Rev. Neurosci. 12, 73–87.
THOMAS P., FENECH M. (2006). A review of genome mutation and Alzheimer’s disease. Mutagenesis 22, 15–33.
TURLEJSKI K. (1996). Evolutionary ancient roles of serotonin: long-lasting regulation of activity and development. Acta Neurobiol. Exp. (Warsz.) 56, 619–636.
UDO H., JIN I., KIM J.-H., LI H.-L., YOUN T., HAWKINS R.D., KANDEL E.R., BAILEY C.H. (2005). Serotonin-Induced Regulation of the Actin Network for Learning-Related Synaptic Growth Requires Cdc42, N-WASP, and PAK in Aplysia Sensory Neurons. Neuron 45, 887–901.
UPTON N., CHUANG T.T., HUNTER A.J., VIRLEY D.J. (2008). 5-HT6 receptor antagonists as novel cognitive enhancing agents for Alzheimer’s disease. Neurotherapeutics 5, 458–469.
VAIDYA V.A., MAREK G.J., AGHAJANIAN G.K., DUMAN R.S. (1997). 5-HT2A receptor-mediated regulation of brain-derived neurotrophic factor mRNA in the hippocampus and the neocortex. J. Neurosci. Off. J. Soc. Neurosci. 17, 2785–2795.
VIALLI M., ERSPAMER V. (1937). Ricerche sul secreto delle cellule enterocromaffini: Nota VII Osservazioni critiche su alcuni problemi inerenti alla istochimica delle enterocromaffini. Z. Für Zellforsch. Mikrosk. Anat. 27, 81–99.
WANG C., YU J.-T., MIAO D., WU Z.-C., TAN M.-S., TAN L. (2013). Targeting the mTOR Signaling Network for Alzheimer’s Disease Therapy. Mol. Neurobiol. 49, 120–135.
WEINSTEIN D., MAGNUSON D., LEE J. (1996). Altered G-protein coupling of a frontal cortical low affinity [3H]8-hydroxy-N,N-dipropyl-2-aminotetralin serotonergic binding site in Alzheimer’s disease. Behav. Brain Res. 73, 325–329.
WILKINSON D., WINDFELD K., COLDING-JØRGENSEN E. (2014). Safety and efficacy of idalopirdine, a 5-HT6 receptor antagonist, in patients with moderate Alzheimer’s disease (LADDER): a randomised, double-blind, placebo-controlled phase 2 trial. Lancet Neurol. 13, 1092–1099.
WOODS Y.L., COHEN P., BECKER W., JAKES R., GOEDERT M., WANG X., PROUD C.G. (2001). The kinase DYRK phosphorylates protein-synthesis initiation factor eIF2Bepsilon at Ser539 and the microtubule-associated protein tau at Thr212: potential role for DYRK as a glycogen synthase kinase 3-priming kinase. Biochem. J. 355, 609–615.
YAMAMOTO T., HIRANO A. (1985). Nucleus raphe dorsalis in Alzheimer’s disease: Neurofibrillary tangles and loss of large neurons. Ann. Neurol. 17, 573–577.
YUN H.-M., KIM S., KIM H.-J., KOSTENIS E., KIM J.I., SEONG J.Y., BAIK J.-H., RHIM H. (2007). The novel cellular mechanism of human 5-HT6 receptor through an interaction with Fyn. J. Biol. Chem. 282, 5496–5505.
ZEINSTRA E.M., WILCZAK N., WILSCHUT J.C., GLAZENBURG L., CHESIK D., KROESE F.G.M., DE KEYSER J. (2006). 5HT4 agonists inhibit interferon-gamma-induced MHC class II and B7 costimulatory molecules expression on cultured astrocytes. J. Neuroimmunol. 179, 191–195.
ZWEIG R.M., ROSS C.A., HEDREEN J.C., STEELE C., CARDILLO J.E., WHITEHOUSE P.J., FOLSTEIN M.F., PRICE D.L. (1988). The neuropathology of aminergic nuclei in Alzheimer’s disease. Ann. Neurol. 24, 233–242.